The occupied MOs of hydrogen cyanide are shown next along with the MO energies. Indicate which AOs
Question:
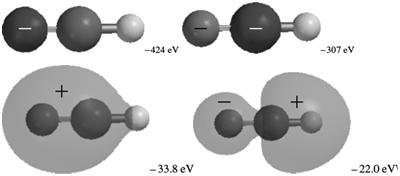
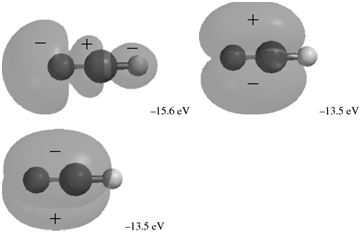
Transcribed Image Text:
-424 ev -307 eV -33.8 eV -22.0 eV メ -15.6 eV -13.5 eV -13.5 eV
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
The MOs corresponding to the energies 424 eV and 307 eV are 1s AOs on N and C respective...View the full answer

Answered By

AJIN kuriakose
I have completed B.Tech in Electrical Engineering & Masters in Power & Control From one of the best universities in India. I got the 99.05 percentile in the Gate Electrical Engineering Exam. I can Help students solving assignments in Electrical subjects like Power Electronics, Control system, Analog, Network Theory & Engineering Mathematics. Clear your fundamentals and develop problem-solving skills and analytical skills to crack the exam.
Get guidance and the opportunity to learn from experienced...
I can provide tuition for Electrical engineering subjects (Power Electronics, Digital electronics, Network Theory, Control System & Engineering Mathematics). The toughest subject of Electrical engineering can be made simple in online classes...
I can also solve it.
1 .I can help you with your assignments or exams or quiz or tutoring.
2. Very strict to the deadlines.
Message me for any help in assignments, live sessions. I am here to help students for all assignments, tests and exams and I will make sure you always get _95% In your subject.
Contact me in solution inn for any help in your semester, projects and for many more things . Also feel free to contact me through solution inn and for any advise related to tutoring and how it works here.thank you.
5.00+
5+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The occupied MOs of ammonia are shown next along with the MO energies. Indicate which AOs are most important in each MO and indicate the relative phases of the AOs. Classify the MOs as localized or...
-
The occupied MOs of ethene are shown next along with the MO energies. Indicate which AOs are most important in each MO and indicate the relative phases of the AOs. Classify the MOs as localized or...
-
Indicate the type of solute-solvent interaction (Section 11.2) that should be most important in each of the following solutions: (a) CCl4 in benzene (C6H6), (b) Methanol (CH3OH) in water, (c) KBr in...
-
2. There are different kinds of statistical analyses, and you will need to use one that is best suited to the data available and the information you need to collect in the given scenario (Good Days)....
-
Determine whether the statement is true or false. Justify your answer. 1. Determine whether the statement is true or false. Justify your answer. In each of the four quadrants, the signs of the secant...
-
The points A and B have coordinates (k, 1) and (8, 2k 1) respectively, where k is a constant. Given that the gradient of AB is 1/3 a. Show that k = 2 b. Find an equation for the line through A and B.
-
Assume two dividend processes Di are independent geometric Brownian motions: dDi Di = i dt + i dBi for constantsi and i and independent Brownian motionsBi. DefineCt = D1t+ D2t. Assume Mt def = et C0...
-
1. Find the optimal March shipment schedule and its total transportation cost for each of the following: a. Cotton b. Polyester c. Silk 2. The company will be opening a silk-making department in the...
-
en TUI come as retirement approaches? 2. Jamie Lee and Ross estimate that they will have $1 million in liquid assets to withdraw from at the start of their retirement. They plan to be in retirement...
-
Which task is not performed by an accountant? A. Monitoring the progress of the business B. Preparing a statement of financial position C. Reporting on the trading results D. Writing up a three...
-
Predict whether the ground state or the first excited state of CH 2 should have the larger bond angle on the basis of the Walsh correlation diagram shown in Figure 24.11. Explain your answer. 1b, 17u...
-
Use the geometrical construction shown in Example Problem 24.8 to derive the electron MO levels for cyclobutadiene. What is the total energy of the molecule? How many unpaired electrons will the...
-
FICA-Medicare Payable is an asset for the employer. True/False LO.1
-
Moving Inc. wants to develop an activity flexible budget for the activity of moving materials. Moving Inc. uses forklifts to move materials from receiving to storeroom and then to production. The...
-
We are in the tail end of Quarter 3 earnings reporting season in the U.S. markets. Roughly 60 percent of companies that have reported their Q3 earnings so far have reported negative earnings relative...
-
Below is a running shock tube illustration. 0.1 0.0 | 0.0 4 4 Diaphragm 1 0.5 Image: Shock tube Initial setup 1 3 2 1 Expansion Head Expansion Tail Slip Shock Surface Image: Running Shock Tube...
-
As you may remember, Holiday Tree Services, Inc. (HTS) has recently entered into a contract with Delish Burger (Delish), whereby HTS is to supply and decorate a Christmas tree in each of Delish...
-
Understanding various types of leadership styles is important in order to determine personal leadership styles. Reflection: Answer both Compare and contrast 2 leadership styles. State the...
-
In Exercises 1 through 28, differentiate the given function. y = 9/t
-
Write a program to move a signed number from smaller register to bigger register. Hint: movzx ax, bl Topic: Data Related Operators and Directives in assembly language
-
At 518C, the rate of decomposition of a sample of gaseous acetaldehyde, initially at a pressure of 363 Torr, was 1.07 Torr s 1 when 5.0 per cent had reacted and 0.76 Torr s 1 when 20.0 per cent had...
-
The ClO radical decays rapidly by way of the reaction, 2 ClO Cl 2 + O 2 . The following data have been obtained: Determine the rate constant of the reaction and the half-life of a ClO radical....
-
The rate law for the reaction in Exercise 22.2a was reported as d[C]/dt = k[A][B][C]. Express the rate law in terms of the reaction rate; what are the units for k in each case? Data in Exercise 22.2a...
-
Assignment Title: The Role of Bookkeeping in Business Management and Financial Reporting Objective: Understand the importance of proper bookkeeping procedures in the management of...
-
17) The adjustment that is made to allocate the cost of a building over its expected life is called:A) depreciation expense.B) residual value.C) accumulated depreciation.D) None of the above answers...
-
9) Prepaid Rent is considered to be a(n):A) liability.B) asset.C) contra-asset.D) expense.10) As Prepaid Rent is used, it becomes a(n):A) liability.B) expense. C) contra-asset.D) contra-revenue.11)...

Study smarter with the SolutionInn App


