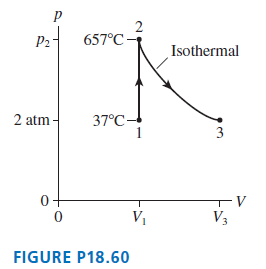
8.0 g of helium gas follows the process 1 ?? 2 ?? 3 shown in FIGURE P18.60.
Question:
8.0 g of helium gas follows the process 1 ?? 2 ?? 3 shown in FIGURE P18.60. Find the values of V1, V3, p2, and T3.
Transcribed Image Text:
P2- 657°C - Isothermal 2 atm - 37°C- 3 0+ -v V1 V3 FIGURE P18.60
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
Model Assume that the helium gas is an ideal gas Visualize Process 1 2 is isochoric proc...View the full answer

Answered By

Fahmin Arakkal
Tutoring and Contributing expert question and answers to teachers and students.
Primarily oversees the Heat and Mass Transfer contents presented on websites and blogs.
Responsible for Creating, Editing, Updating all contents related Chemical Engineering in
latex language
4.40+
8+ Reviews
22+ Question Solved
Related Book For 

Physics for Scientists and Engineers A Strategic Approach with Modern Physics
ISBN: 978-0133942651
4th edition
Authors: Randall D. Knight
Question Posted:
Students also viewed these Physics questions
-
Consider the following gas samples: 4.0 g of hydrogen gas, 4.0 g of helium gas, 1.0 mole of fluorine gas, 44.0 g of carbon dioxide gas, and 146 g of sulfur hexafluoride gas. Arrange the gas samples...
-
A monatomic gas follows the process 1 2 3 shown in Figure EX19.32. How much heat is needed for (a) Process 1 2 (b) Process 2 3? p (atm) 3- 2- 100C isotherm 3 -V (cm) 300 100 200 FIGURE EX19.32
-
Consider a container like that shown in Figure 20.12, with n 1 moles of a monatomic gas on one side and n 2 moles of a diatomic gas on the other. The monatomic gas has initial temperature T 1i ....
-
Court Casuals has 100,000 shares of common stock outstanding as of the beginning of the year and has the following transactions affecting stockholders' equity during the year. May 18 Issues 25,000...
-
Mauve, Inc., uses a two- to six-year graded vesting approach in its retirement plan. Calculate the nonforfeitable percentage for each of the following participants based upon the years of service...
-
Explain how the budgeting process is accomplished in QuickBooks Accountant.
-
What are information quality and environmental quality? Are they related? Why is low-quality information said to drive out high-quality information?
-
Selected transactions for Arnett Advertising Company, Inc., are listed here. 1. Issued common stock to investors in exchange for cash received from investors. 2. Paid monthly rent. 3. Received cash...
-
The company Toyota, SA, offers its employees dental care insurance. A recent study carried out by the director of human resources shows that the annual cost per employee had a normal probability...
-
Consider the following relation: TRIP (Trip_id, Start_date, Cities_visited, Cards_used) This relation refers to business trips made by company salespeople. Suppose the TRIP has a single Start_date...
-
Texas Instruments (TI) designs and manufactures semiconductor products for use in computers, telecommunications equipment, automobiles, and other electronics-based products. The manufacturing of...
-
JCPenney operates a chain of retail department stores, selling apparel, shoes, jewelry, and home furnishings. It also offers most of its products through catalog distribution. During fiscal Year 5,...
-
Evaluate the integrals in Problem. 6x 2 (7 + 2x 3 ) 9 dx
-
How do I view vector and bitmap images in a VR headset?
-
How does PostgreSQL support the evolution of database schemas while preserving data integrity constraints, including techniques such as ALTER TABLE operations, constraint migrations, and backward...
-
How does data replication serve as a cornerstone in distributed computing paradigms, and what are the underlying principles guiding its implementation to ensure fault tolerance, scalability, and data...
-
How does PostgreSQL handle JSON and JSONB data types, and what are the benefits and trade-offs compared to traditional relational data modeling, especially in scenarios involving complex or...
-
My question is how often will a communication satellite needs to communicate with ground control facilities. My question is it often communicating with ground control because it is providing network...
-
What is an example of a metal produced primarily by electrolysis?
-
Les has collected stamps in his spare time for years. He purchased many of his stamps at a price much lower than the current market value. Les recently lost his job as a carpenter. Since his wife...
-
In which of the four basic interactions does an electron participate? A neutrino? A proton? A photon?
-
What is a quark? How many different types of quarks are now known? What are some of the basic properties that distinguish these quarks?
-
Describe the kinds of evidence that have led scientists to conclude that quarks exist.
-
Design an advertising campaign for your virtual business with detail.Virtual Marketing Firm
-
Hayley purchased 9 0 0 shares of Pelican Town Inc. common stock exactly one year ago for $ 5 6 . 6 0 per share. Today, the corporation\'s shares are selling for $ 5 3 . 8 0 per share. Over the last...
-
Games-R-Us is a retail merchandiser that buys from wholesalers and marks-up the price of products (in order to generate a profit). Sales are budgeted at $100,000 for July and $130,000 for August. The...

Study smarter with the SolutionInn App


