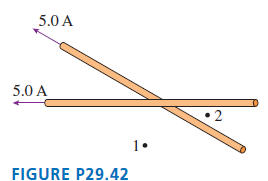
The two insulated wires in FIGURE P29.42 cross at a 30° angle but do not make electrical
Question:
Transcribed Image Text:
5.0 A 5.0 A 1. FIGURE P29.42
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 46% (15 reviews)
Model Assume that the wires are infinitely long and that the magnetic field is ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Physics for Scientists and Engineers A Strategic Approach with Modern Physics
ISBN: 978-0133942651
4th edition
Authors: Randall D. Knight
Question Posted:
Students also viewed these Physics questions
-
Two insulated wires, each 2.40 m long, are taped together to form a two-wire unit that is 2.40 m long. One wire carries a current of 7.00 A; the other carries a smaller current I in the opposite...
-
Two insulated wires perpendicular to each other in the same plane carry currents as shown in Fig. E28.29. Find the magnitude of the net magnetic field these wires produce at points P and Q if the...
-
Two long insulated wires lie in the same horizontal plane. A current of 20.0 A flows toward the north in wire A and a current of 10.0 A flows toward the east in wire B. What is the magnitude and...
-
a. Over a two year time horizon an investor experiences the following net cash flows: Year 0 1 2 Cash flow -$8,000 $5,280 $5,320 What is the internal rate of return for the investor? Demonstrate that...
-
Consider the Bohr model as applied to the following three atoms: (A) neutral hydrogen in the state n = 2; (B) singly ionized helium in the state n = 1; (C) doubly ionized lithium in the state n = 3....
-
Main Street Mullins Sells its muffins to restaurants and coffee houses for an average saling price of 535 per case the following information relates to the budget for Main Street Muffies for this...
-
Download an Excel template for this problem online in MyAccountingLab or at http://www.pearsonhighered.com/Horngren. The James Island Clothing Company began operations on July 1, 2018. The adjusted...
-
A 500-W heating coil designed to operate from 110 V is made of Nichrome wire 0.500 mm in diameter. (a) Assuming that the resistivity of the Nichrome remains constant at its 20.0C value, find the...
-
What are the key challenges in ensuring tax compliance for businesses? How do tax authorities enforce compliance?
-
Avery Company has two divisions, Polk and Bishop. Polk produces an item that Bishop could use in its production. Bishop currently is purchasing 26,000 units from an outside supplier for $16 per unit....
-
A long wire carrying a 5.0 A current perpendicular to the xy-plane intersects the x-axis at x = -2.0 cm. A second, parallel wire carrying a 3.0 A current intersects the x-axis at x = +2.0 cm. At what...
-
What are the strength and direction of the magnetic field at the center of the loop in FIGURE P29.43? 5.0 A 2.0 cm FIGURE P29.43
-
The following incomplete balance sheet for the Sanderson Manufacturing Company was prepared by the company?s controller. As accounting manager for Sanderson, you are attempting to reconstruct and...
-
Begin by locating a scholarly article regarding the effects of increases on minimum wages and discuss its findings. Does a minimum wage increase lead to reductions in employment, or is the overall...
-
Pick an organization (or you can use an example that has professional relevance), and use the DAC framework to accomplish the following objectives. Provide background and a problem statement of the...
-
Woodruff Company is currently producing a snowmobile that uses five specialized parts. Engineering has proposed replacing these specialized parts with commodity parts, which will cost less and can be...
-
What was Newell's organizational culture like before acquiring Rubbermaid? What was the quadrant? Is the perspective short or long term, inside or outside focused, interested in people, process, or...
-
(7.47 of Koretsky 2nd edition) A binary mixture of species 1 and 2 can be described by the following equation of state: P = a 0 VoT The pure species coefficients are given by: amix y1a1a2Y2 with the...
-
Without a calculator, find the exact function value. cos 45
-
Decades after the event, Johnson & Johnson (J&J), the 130-year-old American multinational, is still praised for swiftly The company indicated that its response was based on the expectations set forth...
-
While you are hiking in the wilderness, you find a shiny, glassy-looking mineral. What physical test could you use to determine whether this mineral is a diamond?
-
What makes gold so soft (easily scratched) while quartz and diamond are so much harder?
-
Imagine that we have a liquid with a density of 3.5 g/cm 3 . Knowing that objects of higher density will sink in the liquid, will a piece of quartz sink or float in the liquid? How about a piece of...
-
On consolidated financial statements, where does the parents equity in the net income of the subsidiary account appear? A. On the consolidated income statement, as a revenue B. On the consolidated...
-
Which of the following is not one of the elements of the balanced scorecard? a.cost system b.strategic initiatives c.performance targets d.strategy maps
-
Yield to Maturity and Call with Semiannual Payments Shares Remaining After Recapitalization Dye Trucking raised $75 million in new debt and used this to buy back stock. After the recap, Dye's stock...

Study smarter with the SolutionInn App


