Find the concentrations of the major species in a saturated aqueous solution of LiF. Consider these reactions:
Question:
Find the concentrations of the major species in a saturated aqueous solution of LiF. Consider these reactions:
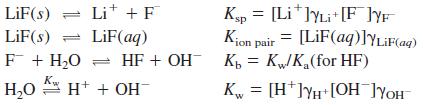
(a) Look up the equilibrium constants in the appendixes and write their pK values. The ion pair reaction is the sum of LiF(s) ⇌Li+ + F- from Appendix F and Li+ + F- ⇋ LiF(aq) from Appendix J. Write the equilibrium constant expressions and the charge and mass balances.
(b) Create a spreadsheet that uses activities to fi nd the concentrations of all species and the ionic strength. Use pF and pOH as independent variables to estimate. It does not work to choose pF and pLi because either concentration fixes that of the other through the relation Ksp = [Li+]gLi+[F-]gF-.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Question Posted:





