Calculate the voltage of the cell by considering the following reactions: By the reasoning in Figure 13-8,
Question:
Calculate the voltage of the cell
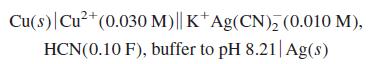
by considering the following reactions:
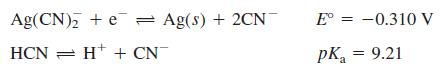
By the reasoning in Figure 13-8, in which direction do electrons flow?
Figure 13-8
Transcribed Image Text:
Cu(s)|Cu²*(0.030 M)|| K†Ag(CN), (0.010 M), HCN(0.10 F), buffer to pH 8.21| Ag(s)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 56% (16 reviews)
To calculate the voltage of the cell in the given electrochemical cell setup we need to identify the ...View the full answer

Answered By

Chandrasekhar Karri
I have tutored students in accounting at the high school and college levels. I have developed strong teaching methods, which allow me to effectively explain complex accounting concepts to students. Additionally, I am committed to helping students reach their academic goals and providing them with the necessary tools to succeed.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Calculate the voltage of the following cell, in which KHP is potassium hydrogen phthalate, the monopotassium salt of phthalic acid. By the reasoning in Figure 13-8, in which direction do electrons...
-
Including activities, calculate the voltage of the cell Ni(s) | NiSO4(0.0020 M) || CuCl2(0.003 0 M)| Cu(s). Assume that the salts are completely dissociated (that is, neglect ion-pair formation). By...
-
Calculate the voltage of each of the following cells. With the reasoning in Figure 13-8, state the direction of electron flow. (a) Fe(s) | FeBr 2 (0.010 M) || NaBr(0.050 M) | Br 2 (l) | Pt(s) (b)...
-
1. Matching Match the graph of the rational function f (x) = (ax + b) / (cx + d) with the given conditions. (a) (b) (c) (d) (i) a > 0 (ii) a > 0 (iii) a 0 b 0 b > 0 b c > 0 c 0 c > 0 d 0
-
Independent random samples of sizes nA = 4 and nB = 2 are taken from two continuous populations. (a) Enumerate all possible collections of ranks associated with the smaller sample in the combined...
-
Presented below are certain operating data for the four departments of Modern Manufacturing Company. Allocate, to the two production departments, the costs of service departments 1 and 2, using...
-
Convert \(2.71 \overline{94}\) to fraction form.
-
Comprehensive Accounting Change and Error Analysis Problem Botticelli Inc. were organized in late 2008 to manufacture and sell hosiery. At the end of its fourth year of operation, the company has...
-
. Quality Office Systems uses a perpetual inventory system. The adjusted trial balance of Quality Office Systems at March 31, 2024, follows: (Click the icon to view the adjusted trial balance.)...
-
Figure P8.31 shows a proposed foundation site, with 10 ft of sand overlying 15 ft of clay with consolidation properties shown. The clay is normally consolidated. Assume 1-D conditions. (a) Compute...
-
(a) Using Reactions 2 and 3, calculate E and write the Nernst equation for the cell. (b) Use the value of K sp for A gI to compute [Ag + ] and find the cell voltage. By the reasoning in Figure 13-8,...
-
(a) Write a balanced equation for the reaction and calculate E for the reaction. (b) Predict whether an equimolar mixture of PuO 2 2 + and PuO + 2 will oxidize H 2 O to O 2 at a pH of 2.00 and P O2 =...
-
Derive an expression for the relationship between pK a and pK b for a conjugate acidbase pair. (pK = -log K.)
-
A company gives the following information: Margin of Safety =3,75,000 Total Cost= 3,87,500 Margin of Safety (Qty.)=15,000 units Break Even Sales in Units=5,000 units Calculate: Selling price per unit...
-
Study the Public Procurement and Disposal of Public Property (Procurement Of Consultants) Regulations, 2021. Respond to the following question based on this legislation. 1. The legislation has eleven...
-
Crab and Apple Co expects that next year's earnings will be eight dollars per share (EPS1 = $8). The company normally pays out 80% of its earnings in dividends to its shareholders and plans to...
-
The following information is provided for each Investment Center Investment Center Cameras Phones Computers Income $ 7,000,000 2,506,000 1,150,000 Average Assets $ 20,900,000 17,900,000 11,400,000...
-
Will the new compensation plan contribute to achieving Dr. Doherty's objectives for the Department of Surgery? Why or why not? What characteristics of the Department of Surgery do you think will...
-
Frannie's Florist produces floral bouquets for commercial businesses (i.e. hotels) and uses an activity-based costing system. Data concerning overhead costs and activity pools is as follows: Activity...
-
If the annual fixed costs are 54,000 dinars, the occupation expense represents 20%, the contribution margin is 25%, and the unit selling price is 40 dinars. Required: Calculate the closing point of...
-
Why does a thermal conductivity detector respond to all analytes except the carrier gas? Why isn't the flame ionization detector universal?
-
Explain what is displayed in a reconstructed total ion chromatogram, selected ion monitoring, and selected reaction monitoring. Which technique is most selective and which is least selective and why?
-
Use Table 23-3 to predict the elution order of the following compounds from columns containing (a) poly(dimethylsiloxane),(b) (diphenyl)0.35(dimethyl)0.65polysiloxane, and (c) poly(ethylene glycol):...
-
3 B B B B B B B B B ORTHOGRAPHIC NAME: PROJECTION CHAPTER 02 EXERCISE 11 DATE: VISUALIZATION & ENGINEERING DESIGN GRAPHICS WITH AUGMENTED REALITY SKETCH ALL NECESSARY VIEWS TO COMPLETELY DESCRIBE THE...
-
You are about to optimize the IP addressing table for a network with 5 different subnetworks and 20 hosts pers subnetwork. The Internet Service Provider gives you a class B network. The best...
-
%m-file to calculate eigenvalues and eigenvectors % inertia values in kg-m^2 J1=5.65; J2=15.6; J3=16.38; J4=20.45; J5=29.38; J6 .5*15818; % stiffness values in N-m/rad K1=1.7e06; K2 3.4e06;...

Study smarter with the SolutionInn App


