Answered step by step
Verified Expert Solution
Question
1 Approved Answer
10.1 The formal charge is the charge an element would have in a molecule or ion if all of the bonding electrons were shared equally
10.1 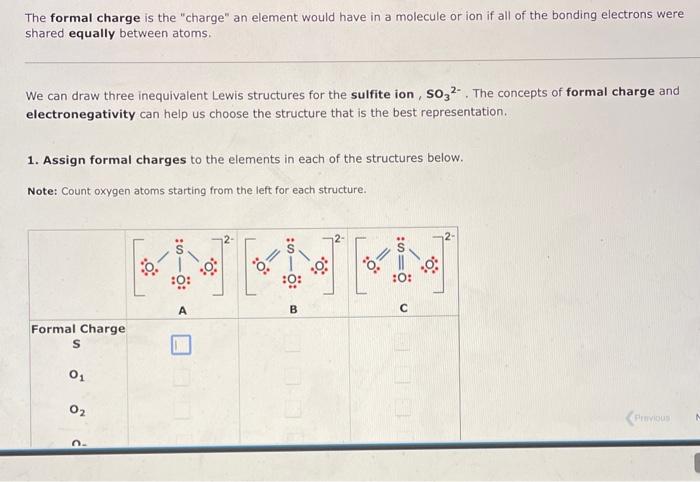
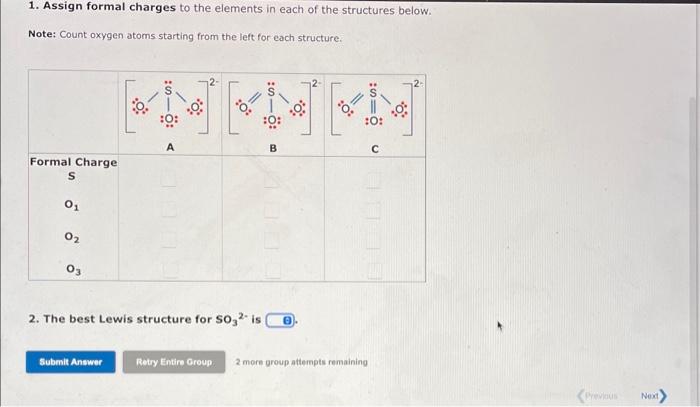
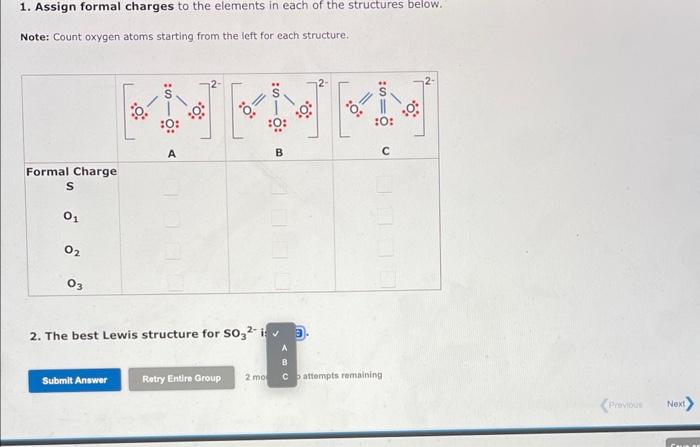
The formal charge is the "charge" an element would have in a molecule or ion if all of the bonding electrons were shared equally between atoms. We can draw three inequivalent Lewis structures for the sulfite ion, SO32. The concepts of formal charge and electronegativity can help us choose the structure that is the best representation. 1. Assign formal charges to the elements in each of the structures below. Note: Count oxygen atoms starting from the left for each structure. 1. Assign formal charges to the elements in each of the structures below. Note: Count oxygen atoms starting from the left for each structure. 2. The best Lewis structure for SO32 is 1. Assign formal charges to the elements in each of the structures below. Note: Count oxygen atoms starting from the left for each structure. 2. The best Lewis structure for SO32 


Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


