Answered step by step
Verified Expert Solution
Question
1 Approved Answer
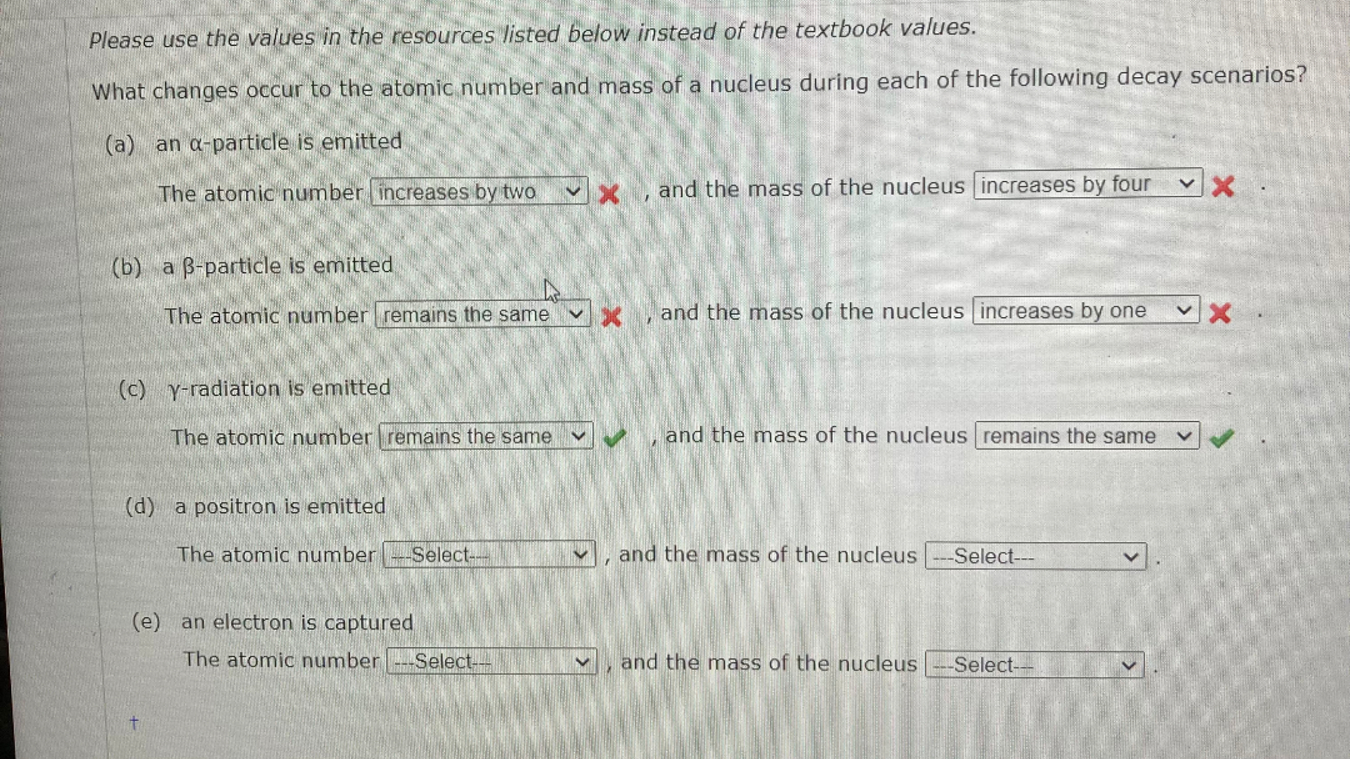
Please use the values in the resources listed below instead of the textbook values. What changes occur to the atomic number and mass of a
Please use the values in the resources listed below instead of the textbook values.
What changes occur to the atomic number and mass of a nucleus during each of the following decay scenarios?
a an particle is emitted
The atomic number increases by two and the mass of the nucleus increases by four
b a particle is emitted
The atomic number remains the same and the mass of the nucleus increases by one
cradiation is emitted
The atomic number
and the mass of the nucleus
d a positron is emitted
The atomic number
and the mass of the nucleus
e an electron is captured
The atomic number
and the mass of the nucleus
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


