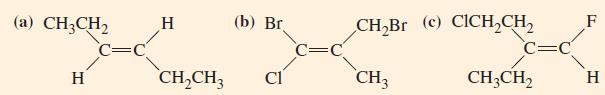
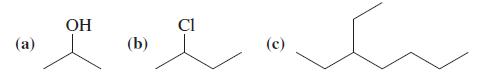
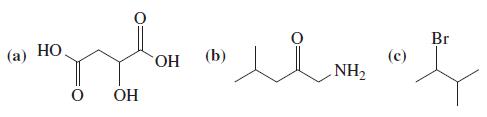
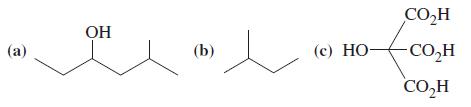
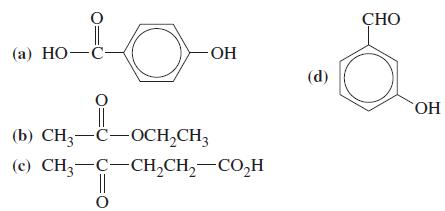
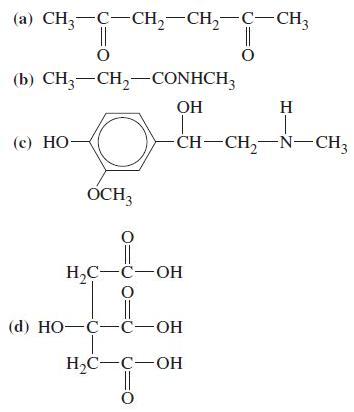
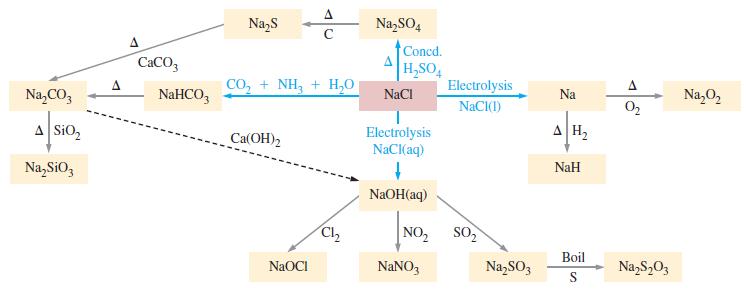
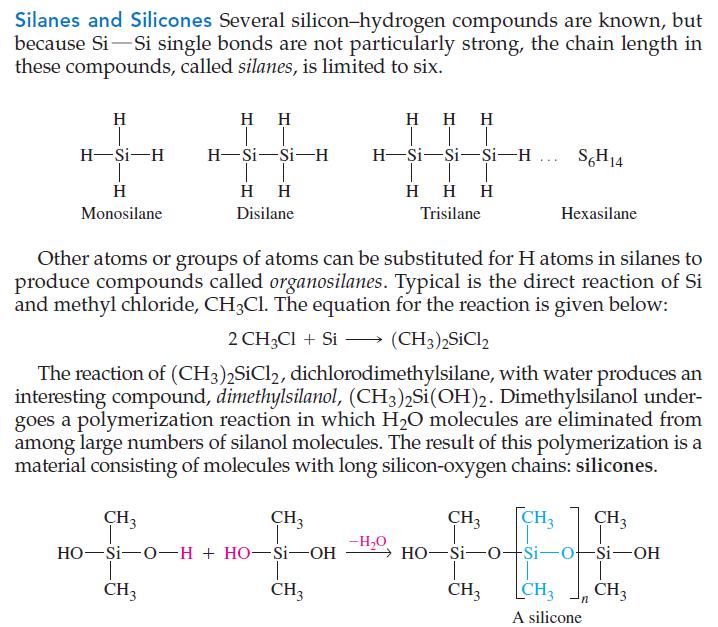
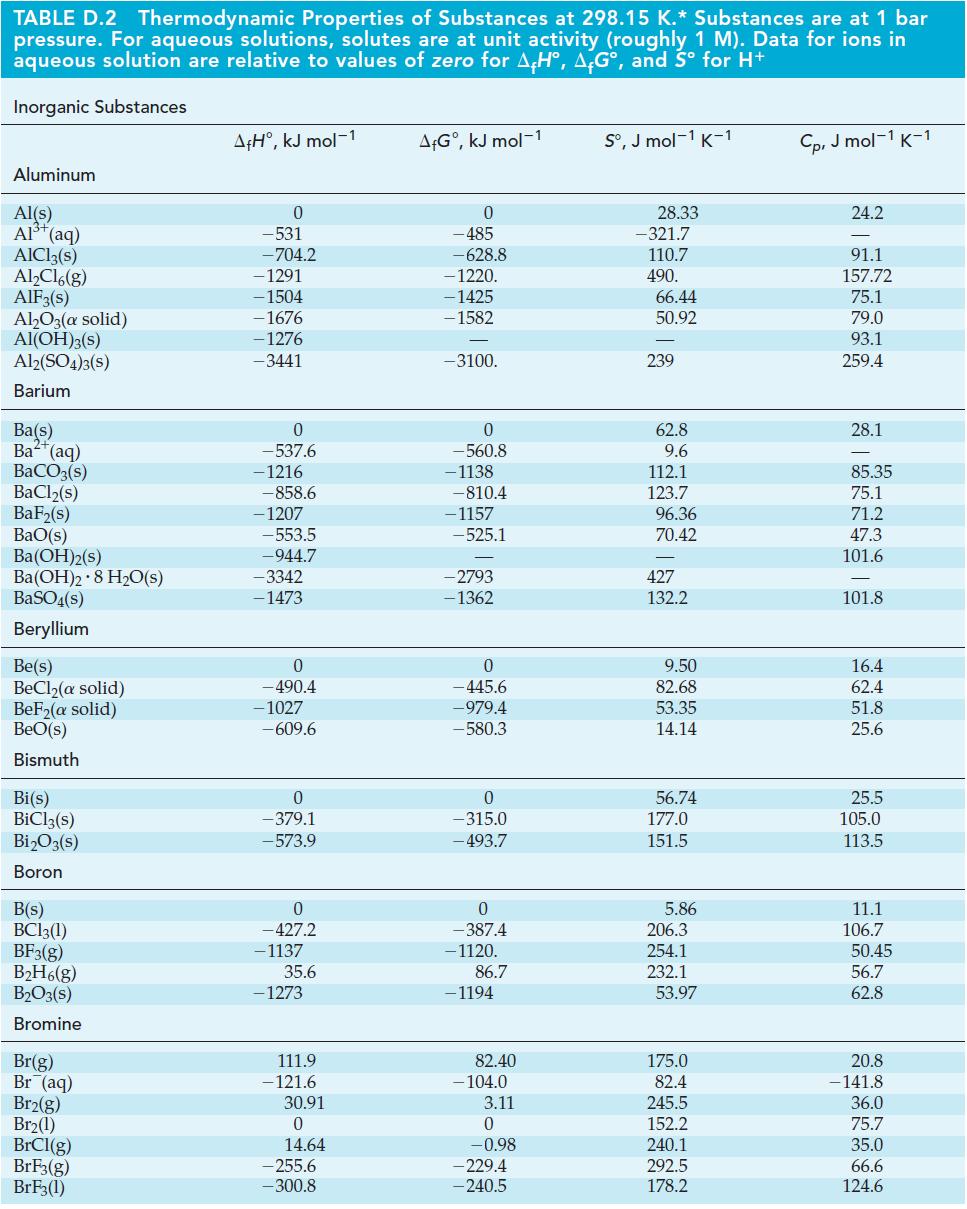
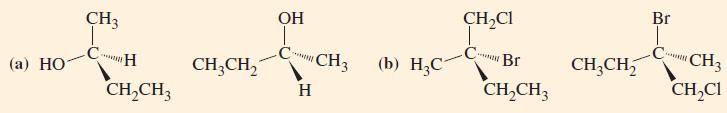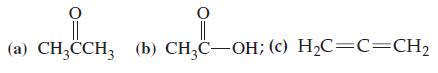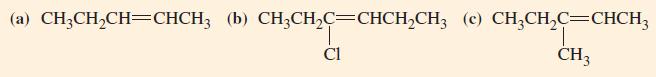General Chemistry Principles And Modern Applications 11th Edition Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette - Solutions
Unlock your understanding of chemistry with our comprehensive resources for "General Chemistry: Principles And Modern Applications, 11th Edition" by Ralph Petrucci and others. Access the complete solution manual and answers key online, providing you with step-by-step solutions and solved problems that enhance your learning experience. Dive into chapter solutions and test banks, tailored to guide you through textbook questions and answers with ease. Whether you need an instructor manual or solutions PDF, our platform offers everything you need for academic success. Explore our resources for free download and elevate your chemistry knowledge today!
![]()
![]() New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
![]()
![]()


![Acidic solution ([H+] = 1 M): +7 +5 CIO4 1.189 V CIO4 CIO3 1 I 0.374 V Basic solution ([OH-] = 1 M): +7 +5](https://dsd5zvtm8ll6.cloudfront.net/images/question_images/1701/2/4/1/7556566e39b91d421701241752935.jpg)

![Acidic solution ([H+] = 1 M): +7 +5 1.189 V CIO4- CIO4 CIO3 0.374 V I Basic solution ([OH-] = 1 M): +7 +5](https://dsd5zvtm8ll6.cloudfront.net/images/question_images/1701/2/4/3/5596566eaa7878f41701243559378.jpg)