Propose efficient methods for accomplishing each of the following transformations. Most will require more than one step.
Question:
Propose efficient methods for accomplishing each of the following transformations. Most will require more than one step.
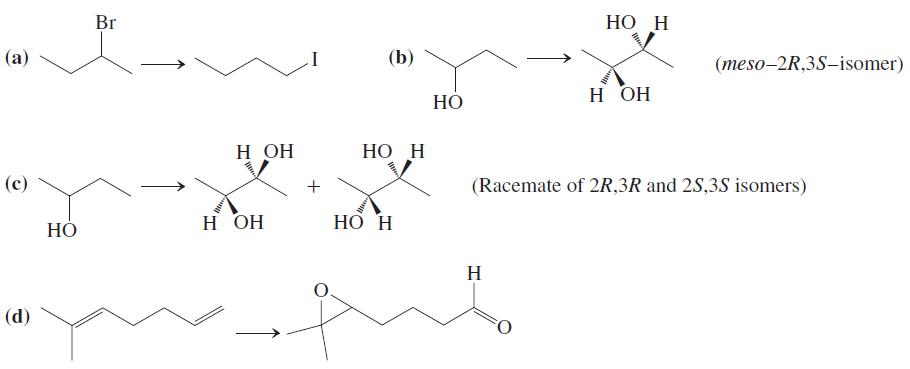
Transcribed Image Text:
Br НО Н (а) (b) (meso-2R,3S–isomer) н он НО Н ОН НО Н (с) (Racemate of 2R,3R and 2S,3S isomers) н он Hồ H НО НО Н H (d)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
a b c d ...View the full answer

Answered By

Nikhil Sharma
I am teaching for 4 years and as a result of that I have very clear idea about the student's difficulties and I know how to rectify them. I have deep knowledge of my subject as I have done Masters in it for finest institute of the country.
I have cleared various nation level competitive exams with very good ranks so I can help students to develop competitive attitude.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Show how you would accomplish the following transformations. Some of these examples require more than one step. (a) 2-methylpropene 2, 2-dimethyloxirane (b) 1-phenylethanol 2-phenyloxirane (c)...
-
BACKGROUND You are an information analyst working for NEE. The company president has asked you to prepare a Quantitative analysis of financial, sales, and operations data to help determine which...
-
Each of the following scenarios requires the use of accounting information to carry out one or more of the following managerial activities: (1) Planning, (2) Control and evaluation, (3) Continuous...
-
Antonio Banderos & Scarves makes headwear that is very popular in the fall/winter season. Units sold are anticipated as: October .......... 1,250 November ......... 2,250 December ......... 4,500...
-
Describe the key differences between doing a qualitative risk assessment versus a quantitative risk assessment.
-
A monophyletic group is a ________. a. Phylogenetic tree b. Shared derived trait c. Character state d. Clade
-
Suggest particular ethical questions facing HRM people in the future? LO1
-
Tom Reynolds is the sole proprietor of Pampered Pets, a business specializing in the sale of high- end pet gifts and accessories. Pampered Pets sales totaled $ 986,000 during the most recent year....
-
The following information was extracted from the Books of Anderson for the year ended 30 June 2021. Sales Ledger Balances at 01 July 2020 Sales Ledger Balances at 30 June 2021 John Moore Rs 2 300...
-
Balance a ration for a milking dairy cow to contain: CP = 16 % Ca = .66 % P= .41 % 27 % Corn Silage Feed Feed Feed 23 % Alfalfa 5 % Brewers Grains Feed 3 % Whole Cotton Seed Leave 2% slack space for...
-
Show how you would synthesize each of the following molecules from an alkene of appropriate structure (your choice). Br H () (b) I (meso-4R,5S-isomer) H Br H Br Br H (d) (Racemate of 4R,5R and 4S,5S...
-
Without consulting the Reaction Road Map on pp. 556 557, suggest a reagent to convert a general alkene, into each of the following types of compounds. H C=C
-
Describe a consignment sale. When does a consignor recognize revenue for a consignment sale?
-
P14-26. Forecasting with Parsimonious Method and Estimating Share Value Using the ROPI Model Following are income statements and balance sheets for Cisco Systems. CISCO SYSTEMS Consolidated...
-
A little lesson on horseracing.An exacta wager is where you pick the horse that you think will come first, and another who will come second. A trifecta wager is where you pick 3 horses that you think...
-
Avery, an unmarried taxpayer, had the following income items: Salary Net income from a rental house 3 7 , 0 5 0 4 , 9 0 0 Avery has a 4 - year - old child who attends a child care center. Assume the...
-
California Lottery Let A denote the event of placing a $1 straight bet on the California Daily 4 lottery and winning. There are 10,000 different ways that you can select the four digits (with...
-
"Tamara Wiley glanced in the mirror before leaving her apartment and heading to her 8 a.m. class. She was having a bad hair day, so she had thrown on a scarf. Her quick check in the mirror told her...
-
Write a piece of code that reads a shorthand text description of a playing card and prints the longhand equivalent. The shorthand description is the cards rank ( 2 through 10, J, Q, K, or A )...
-
You are a U.S. investor who purchased British securities for 2,000 one year ago when the British pound cost U.S. $1.50. What is your total return (based on U.S. dollars) if the value of the...
-
The unfolding, or denaturation, of a biological macromolecule may be brought about by treatment with substances, called denaturants that disrupt the intermolecular interactions responsible for the...
-
The compound p-azoxyanisole forms a liquid crystal. 5.0 g of the solid was placed in a tube, which was then evacuated and sealed. Use the phase rule to prove that the solid will melt at a definite...
-
Use a phase diagram like that shown in Fig. 6.36 to indicate how zone leveling may be described.
-
Lambert bought a second-hand car from his local dealership. Before agreeing to buy the car, the salesperson told Lambert that the car costs only $200, because it was nearly 23 years old and very...
-
Inventory analysis A company reports the following: Line Item DescriptionAmountCost of goods sold$696,000Average inventory58,000 Determine (a) the inventory turnover and (b) the days' sales in...
-
Hedging should not be treated as an additional source of profit because A. your losses on one side should about equal your gains on the other side. B. you should try to make money on both sides of...

Study smarter with the SolutionInn App


