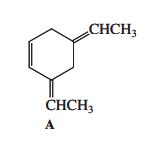
When compound A (shown in the margin) is treated with dilute mineral acid, an isomerization takes place.
Question:
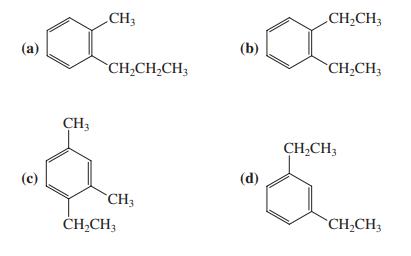
When compound A (shown in the margin) is treated with dilute mineral acid, an isomerization takes place. Which of the following compounds is the new isomer formed?
Transcribed Image Text:
CHCH3 CHCH3 A
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
CH CH3 CHCH3 d CH ...View the full answer

Answered By

Md Abu Zaid
I started my college journey since2011. I did my intermediate from government tirhut higher secondary school Mehsi in 2012, then I did complete my graduation in 2017 from Langat Singh college Muzaffarpur in Chemistry. After that I joined Indian Institute of Technology Bhubaneswar in 2017 and i completed my masters in 2019. Actually I have been teaching since 2017. I taught chemistry as well as mathematics because, I was very interested in science since childhood.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
When cyclooctyne is treated with dilute, aqueous sulfuric acid and HgSO4, a new compound results. It is best represented as () (b) () OH OH (d)
-
Vanillin, whose structure is shown in the margin and is the subject of the Chapter Opening, is a benzene derivative with several functional groups, each one of which displays its characteristic...
-
Structure A (shown in the margin) is that of a naturally occurring a-amino acid. Select its name from the following list. (a) Glycine; (b) Alanine; (c) Tyrosine; (d) Cysteine. H,N- -H- CH3 A
-
In the Walton Bookstore example with a discrete demand distribution, explain why an order quantity other than one of the possible demands cannot maximize the expected profit.
-
What is the fourth constraint of project management that some authors include?
-
What is a harmonic function? A harmonic conjugate?
-
Explain the framework of criminal law in the health and safety field and its enforcement? LO1
-
Education reform is one of the most hotly debated subjects on both state and national policy makers list of socioeconomic topics. Consider a linear regression model that relates school expenditures...
-
Imagine a situation where the deposits at state-chartered banks would be insured by a state insurance fund and deposits at nationally chartered banks would be insured by FDIC. How would you expect...
-
Given the following product tree, complete the MRP records for parts X, Y, W, and Z. Note that parts X and Y have specified order quantities. Z(1) Y(3) W(1) X Z(2) Part X Lead Time: 1 week Lot Size:...
-
The species that is not aromatic according to Hckels rule is () (b) () (d)
-
Which set of reagents will best carry out the conversion shown? (a) HBr, peroxides; (b) Br2, FeBr3; (c) Br2 in CCl4; (d) KBr Br
-
Briefly describe the typical steps in the typical systems development life cycle (SDLC) as presented in this chapter.
-
Required information [The following information applies to the questions displayed below.] Victory Company uses weighted-average process costing to account for its production costs. Conversion cost...
-
Finer, % 100 90 80 70 60 50 40 30 20 10 0 0.01 0.1 1 Size, mm L 10 100 Figure shows a grain size distribution curve of soil. Estimate the coefficient of curvature (Cc) of this soil.
-
Follows is a list of outstanding invoices at 12/31/09. List is by customer. Company: Winter Invoice: 101 Date: Amount: 4/15 300.00 155 7/1 500.00 162 10/14 600.00 197 12/16 250.00 Bradley 126 6/25...
-
Question 3. The acceleration of a robot as it moves along a straight line in the horizontal x-axis is given by -kt a = e (2 cos wt +3 sin wt), k = 0, w % 0, where k and w are positive constants and...
-
(1 pt) To find the length of the curve defined by from the point (0,0) to the point (1,9), you'd have to compute where a b= and f(x)= y=5x+4x / f(x)dx
-
Selected data from Emporia Company follow. Required Compute the following and round computations to two decimal points: a. The accounts receivable turnover for Year 3. b. The inventory turnover for...
-
Cleaning Service Company's Trial Balance on December 31, 2020 is as follows: Account name Debit Credit Cash 700 Supplies Pre-paid insurance Pre-paid office rent Equipment Accumulated depreciation -...
-
The coordinates of the four I atoms in the unit cell ofKI04 are (0,0,0), (O, , ), (, , , ), (,O,) By calculating the phase of the I reflection in the structure factor, show that the I atoms...
-
The treatment in Problem 20.28 applies only to one-dimensional solids. In three dimensions, the variation of density of states is more like that shown in Fig. 20.70. Account for the fact that in a...
-
An NO molecule has thermally accessible electronically excited states. It also has an unpaired electron, and so may be expected to be paramagnetic. However, its ground state is not paramagnetic...
-
Randy Corp. issued $ 2 0 0 , 0 0 0 of 7 . 6 % ( payable each 2 8 February and 3 1 August ) , 4 - year bonds. The bonds were dated 1 March 2 0 4 , and mature on 2 8 February 2 0 8 . The bonds were...
-
with showing the steps please...
-
A commercial office lease requires an immediate payment of $24,000 plus $24,000 per year at the end of each of the next 10 years. At a discount rate of 14 percent, what is the present value of this...

Study smarter with the SolutionInn App


