Estimate the c p of nitrogen at 300 kPa and 400 K, using (a) The relation in
Question:
Estimate the cp of nitrogen at 300 kPa and 400 K, using
(a) The relation in Prob. 12–91,
Data From Q#91:
Consider an infinitesimal reversible adiabatic compression or expansion process. By taking s = s(P, v) and using the Maxwell relations, show that for this process Pvk = constant, where k is the isentropic expansion exponent defined as
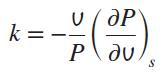
Also, show that the isentropic expansion exponent k reduces to the specific heat ratio cp/cv for an ideal gas.
(b) Its definition. Compare your results to the value listed in Table A–2b.
Transcribed Image Text:
U ( OP k = - P du
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
The c p of nitrogen at 300 kPa and 400 K is to be estimated using the relation given and its defini...View the full answer

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Thermodynamics An Engineering Approach
ISBN: 9781259822674
9th Edition
Authors: Yunus Cengel, Michael Boles, Mehmet Kanoglu
Question Posted:
Students also viewed these Engineering questions
-
Consider an infinitesimal reversible adiabatic compression or expansion process. By taking s = s(P, v) and using the Maxwell relations, show that for this process Pvk = constant, where k is the...
-
Consider an infinitesimal reversible adiabatic compression or expansion process. By taking s = s(P, v) and using the Maxwell relations, show that for this process Pvk = constant, where k is...
-
Estimate the cp of nitrogen at 300 kPa and 400 K, using (a) The relation in the above problem and (b) Its definition. Compare your results to the value listed in Table A2b.
-
Show that for an integer n > 2, the period of the decimal expression for the rational number is at most n - 1. Find the first few values of n for which the period of - is equal ton- 1. Do you notice...
-
The accompanying table lists weights (in kilograms) of plastic discarded by a sample of households, along with the sizes of the households (adapted from Data Set 1). Is there a significant linear...
-
DE19-11 Look at Exhibit 19-11 (page 766). What would be Top-Flight's cost of goods sold and operating income if the cost of goods manufactured were $35.000 rather than $42,000? (Other costs and...
-
What competencies best describe (a) your college or university and (b) your favorite restaurant? LO.1
-
Slick Pads is a company that manufactures laptop notebook computers. The company is considering adding its own line of computer printers as well. It has considered the implications from the marketing...
-
Using one of the framework as you starting point describe how circular economy will influence or impact accounting in the future
-
1. What are some advantages of a person-focused pay system at Mitron? 2. What are some disadvantages? 3. What approach would you recommend for Holly to take in designing a person-focused pay system?...
-
For ideal gases, the development of the constant-volume specific heat yields Prove this by using the definitions of pressure and temperature, T = (u/s)v and P = (u/v)s. = 0 du.
-
Propane at 500 kPa and 100C is compressed in a steady-flow device to 4000 kPa and 500C. Calculate the change in the specific entropy of the propane and the specific work required for this compression...
-
Describe the systems used by various management groups within the firm in terms of the information they use, their outputs, and groups served.
-
(Reference: A Closer Look on Cost Accounting, De Jesus, 2019) Paulo Corporation had the following account balances as of August 1, 2020: Raw materials inventory (direct and indirect) Work in Process...
-
Use the given data values (a sample of female arm circumferences in centimeters) to identify the corresponding z scores that are used for a normal quantile plot, then identify the coordinates of each...
-
1. What stakeholders other than customers, suppliers, and partners should be considered when conceiving ways to monetize data? 2. What would be one strategy to maximize "reliability" attribute in...
-
What important guidance does little's law offer healthcare leadership regarding the planning and utilization of resources
-
Semester Two Practice Examinations, 2022 STAT2201 Question 2. [10 marks] A study investigated the effect of playing computer games on heart rate. Twenty eight individuals were recruited into the...
-
For the part shown, clearly identify each of the following, with labels and sketches on the drawing. (a) Datum features A, B, and C. (b) Datums A, B, and C. (c) Datum reference frame based on datum...
-
When the Department of Homeland Security created a color-coded system to prepare government officials and the public against terrorist attacks, what did it do right and what did it do wrong?
-
A Heat pump water heater (HPWH) heats water by absorbing heat from the ambient air and transferring it to water. The heat pump has a COP of 3.4 and consumes 6 kW of electricity when running....
-
A heat pump that operates on the ideal vapor-compression cycle with refrigerant-134a is used to heat a house. The mass flow rate of the refrigerant is 0.25 kg/s. The condenser and evaporator...
-
A large refrigeration plant is to be maintained at - 15oC, and it requires refrigeration at a rate of 100 kW. The condenser of the plant is to be cooled by liquid water, which experiences a...
-
Arnold inc. is considering a proposal to manufacture high end protein bars used as food supplements by body builders. The project requires an upfront investment into equipment of $1.4 million. This...
-
Billy Bob bank has three assets. It has $83 million invested in consumer loans with a 3-year duration, $46 million invested in T-Bonds with a 12-year duration, and $69 million in 6-month (0.5 years)...
-
Ventaz Corp manufactures small windows for back yard sheds. Historically, its demand has ranged from 30 to 50 windows per day with an average of 4646. Alex is one of the production workers and he...

Study smarter with the SolutionInn App


