Treatment of a carboxylic acid with trifluoroacetic anhydride leads to an unsymmetrical anhydride that rapidly reacts with
Question:
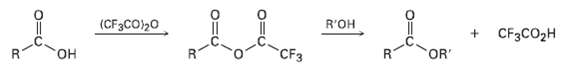
Treatment of a carboxylic acid with trifluoroacetic anhydride leads to an unsymmetrical anhydride that rapidly reacts with alcohol to give an ester.
(a) Propose a mechanism for formation of the unsymmetrical anhydride.
(b) Why is the unsymmetrical anhydride unusually reactive?
(c) Why does the unsymmetrical anhydride react as indicated rather than giving a trifluoroacetate ester plus carboxylicacid?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Question Posted:





