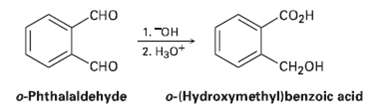
When o-phthalaldehyde is treated with base, o-(hydroxymethyl) benzoic acid is formed. Show the mechanism of this reaction.
Question:
When o-phthalaldehyde is treated with base, o-(hydroxymethyl) benzoic acid is formed. Show the mechanism of this reaction.
Transcribed Image Text:
Сно CO2H 1. "OH 2. Нзо* "сно "CH2он o-Phthalaldehyde o-(Hydroxymethyl)benzoic acid
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 40% (15 reviews)
0 OH addition of OH O COH H 0 H Th...View the full answer

Answered By

Asim farooq
I have done MS finance and expertise in the field of Accounting, finance, cost accounting, security analysis and portfolio management and management, MS office is at my fingertips, I want my client to take advantage of my practical knowledge. I have been mentoring my client on a freelancer website from last two years, Currently I am working in Telecom company as a financial analyst and before that working as an accountant with Pepsi for one year. I also join a nonprofit organization as a finance assistant to my job duties are making payment to client after tax calculation, I have started my professional career from teaching I was teaching to a master's level student for two years in the evening.
My Expert Service
Financial accounting, Financial management, Cost accounting, Human resource management, Business communication and report writing. Financial accounting : • Journal entries • Financial statements including balance sheet, Profit & Loss account, Cash flow statement • Adjustment entries • Ratio analysis • Accounting concepts • Single entry accounting • Double entry accounting • Bills of exchange • Bank reconciliation statements Cost accounting : • Budgeting • Job order costing • Process costing • Cost of goods sold Financial management : • Capital budgeting • Net Present Value (NPV) • Internal Rate of Return (IRR) • Payback period • Discounted cash flows • Financial analysis • Capital assets pricing model • Simple interest, Compound interest & annuities
4.40+
65+ Reviews
86+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show the mechanism of these two reactions. E A or B (5 mol%) CH2CI2, 25 C, 1 hE E. E E = CO,Me 10a (93-98%) 10b A or B (5 mol%) CH,Cl2, 25 C, 1.5 h 11 12 (73-77%)
-
When benzoic acid is allowed to stand in water enriched in 18O, the isotopic label becomes incorporated into the benzoic acid. The reaction is catalyzed by acids. Suggest an explanation for this...
-
When benzene is treated with propene and sulfuric acid (see structures of reactants below), two different monoalkylation products are possible. Draw their structures. Which one do you expect to be...
-
Two kilograms of water, initially saturated liquid at 10 kPa, are heated to saturated vapor while the pressure is maintained constant. Determine the work and the heat transfer for the process, each...
-
During a job interview, Pam Thompson is offered a salary of $28,000. The company gives annual raises of 4 percent. What would be Pam's salary during her fifth year on the job?
-
A judge mandates that juvenile offenders who have priors be assigned to a trained delinquency prevention mentor. To assess this intervention, offenders will be randomly assigned to one of two groups:...
-
Clarify the difference between sample statistics and population parameters
-
Both Waste, Inc. (a private trash collector) and Croton Falls Village will acquire new sanitation trucks. Both will borrow long term, making similar financial arrangements to finance the trucks, and...
-
what is the link between public financial management and financial management systems
-
Projection bias causes people to suppose that dialysis patients have a much lower quality of life than actually prevails. However, when prompted by researchers to think about the ways they will adapt...
-
?-Carotene a yellow food-coloring agent and dietary source of vitamin A, can be prepared by a double Witting reaction between 2 equivalents of ?-ionylideneacetaldehyde and a diylide. Show the...
-
What is the stereochemistry of the pyruvate reduction shown in figure, does NADH lose its pro-R or pro-S hydrogen? Does addition occur to the Si face or Re face of pyruvate?
-
Let x [n] and h [n] be two real finite-length sequences such that x[n] = 0 for n outside the interval 0 ? n ? L ? 1, h[n] = 0 for n outside the interval 0 ? n ? p ? 1. We wish to compute the sequence...
-
1. Write short notes on Discrete Cosine Transform (DCT) 2. Describe Fast Fourier Transform 3. Write short notes on the Hotelling transform
-
1Explain Discrete Fourier Transform in detail. 2. Explain the Properties of 2D discrete Fourier Transform 3. Discuss Hadamard's transformation in detail
-
1. Specify the objective of image enhancement technique. 2. List the 2 categories of image enhancement. 3. What is the purpose of image averaging? 4. What is meant by masking? 5. Define histogram.
-
6. What is meant by histogram equalization? 7. Differentiate linear spatial filter and non-linear spatial filter. 8. Give the mask used for high boost filtering. 9. What is meant by a laplacian...
-
11. What do you mean by Point processing? 12. Define Derivative filter? 13. Define spatial filtering 14. What is a Median filter? 15. What is maximum filter and minimum filter? 16. Write the...
-
Succeeding Through Innovation Synopsis This guide focuses on the key business process of innovation, the exploitation of new ideas, and the factors most relevant to this. The promotion of...
-
CdF2 (s) Cd+ (aq) + 2 F- (aq) 1. A saturated solution of CdF2 is prepared. The equilibrium in the solution is represented above. In the solution [Cd+] eq = 0.0585 M and [F-] eq = 0.117 M. a....
-
Indicate several ways in which the valence bond method is superior to Lewis structures in describing covalent bonds.
-
(R)-(+)-Glyceraldehyde can be transformed into (+)-malic acid by the following synthetic route. Give stereochemical structures for the products of each step. Bra H20 PBra R-+)-Glyceraldehyde...
-
R)-(+)-Glyceraldehyde can also be transformed into (-)-malic acid. This synthesis begins with the conversion of (R)-(+)-glyceraldehyde into (-)-tartaric acid, as shown in Problem 17.41, parts (e) and...
-
Cantharidin is a powerful vesicant that can be isolated from dried beetles (Cantharis vesicatoria, or "Spanish fly"). Outlined here is the stereospecific synthesis of cantharidin reported by Gilbert...
-
Transcribed image text
-
QUESTION 20 Assume a company reported the following results Sales Net operating income Average operating assets Margin Turnover Return on investment (ROI) 5300,000 2 $240.000 40% ? 2 What is the net...
-
2. Using the graph provided below, determine the fixed cost, the total variable cost, the variable cost per unit, and the TOTAL COST to produce 60 units. Fixed Cost ______________ Variable Cost...

Study smarter with the SolutionInn App


