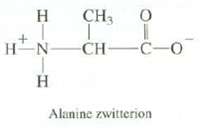
Amino acids such as alanine actually exist as species called zwitterions, with a positive charge on the
Question:
Amino acids such as alanine actually exist as species called zwitterions, with a positive charge on the nitrogen and a negative charge on the oxygen. Explain what effect you expect this to have on the melting point of alanine.
Transcribed Image Text:
CH3 0 T HN CH- HINIH Н Н I -C-0 Alanine zwitterion
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (17 reviews)
The zwitterions form of al...View the full answer

Answered By

Somshukla Chakraborty
I have a teaching experience of more than 4 years by now in diverse subjects like History,Geography,Political Science,Sociology,Business Enterprise,Economics,Environmental Management etc.I teach students from classes 9-12 and undergraduate students.I boards I handle are IB,IGCSE, state boards,ICSE, CBSE.I am passionate about teaching.Full satisfaction of the students is my main goal.
I have completed my graduation and master's in history from Jadavpur University Kolkata,India in 2012 and I have completed my B.Ed from the same University in 2013. I have taught in a reputed school of Kolkata (subjects-History,Geography,Civics,Political Science) from 2014-2016.I worked as a guest lecturer of history in a college of Kolkata for 2 years teaching students of 1st ,2nd and 3rd year. I taught Ancient and Modern Indian history there.I have taught in another school in Mohali,Punjab teaching students from classes 9-12.Presently I am working as an online tutor with concept tutors,Bangalore,India(Carve Niche Pvt.Ltd.) for the last 1year and also have been appointed as an online history tutor by Course Hero(California,U.S) and Vidyalai.com(Chennai,India).
4.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Why are amino acids such as arginine and histidine required in relatively large amounts by children but in smaller amounts by adults? The adult human is not able to make these amino acids.
-
Calculate the formal charge on the nitrogen atom in ammonia, NH3; in the ammonium ion, NH4+; and in the amide ion, NH2.
-
If you painted positive charge on the floor, what surface charge density would be necessary to suspend a 15C, 5.0-g particle above the floor?
-
Consider a soap bubble. Is the pressure inside the bubble higher or lower than the pressure outside?
-
The 'Peal life' suggests that senior executive salaries have risen considerably over time. What do Herzberg's theory of motivation and expectancy theory suggest about using large salary packages to...
-
Green Gooey Company is evaluating all of its internal control processes. Betty Webit, the controller, believes that overall the processes are fairly decent, but she is concerned that there is a lack...
-
Which of the following is not a cash equivalent? a. Short-term U.S. Treasury bill b. Short-term certificate of deposit c. Money-market account d. IBM common stock
-
1. Name three major trade-offs in process selection. 2. What trade- offs are involved when deciding how often to rebalance an assembly line? 3. Who needs to be involved in process selection? 4. Who...
-
FINANCIAL ANALYSIS: Vertical, Horizontal and Ratio Analysis (CP 4A) Compile common size income statement and common size balance sheet (vertical and horizontal analysis) for the 3 years of financial...
-
GENERAL INSTRUCTIONS: You will execute multiple regression models using data in the worksheets, "Time Series Data" and "Cross Sectional Data." For all regressions, you will use the same dependent...
-
Glucose is a typical carbohydrate. What functional groups are present in glucose? What would you predict about the water solubility of glucose, oiler a reason why nature uses carbohydrates rather...
-
What is the DU of estrone, a female sex hormone? Use the number of carbons and oxygen?s in estrone to calculate the number of hydrogen?s it has. Estrone 0 CH3
-
Consider an economy that is populated by a bank, consumers whose population is normalized to be 1, and a regulator. Each consumer is born in \(t=0\) with 1 unit of endowment, and they can invest in...
-
Income Statement Enter the information from the income statement. Total Operating Income (or Profit/Loss) 169,784 Total Operating Revenue 8,155,676 Total Operating Expenses 7,985,892 Net Patient...
-
Explain: 1.How does financial leverage affect ROE and EPS? 2.What are the three capital structure cases?
-
Culver Company lost most of its inventory in a fire in December just before the year-end physical inventory was taken. Corporate records disclose the following. Inventory (beginning) $80,000 Sales...
-
Double West Suppliers (DWS) reported sales for the year of $280,000, all on credit. The average gross profit percentage was 40 percent on sales. Account balances follow: Accounts receivable (net)...
-
- Job 652 was recently completed. Manufacturing overhead is allocated based on direct labor hours (DLH). The following data have been recorded on its job cost sheet: Direct materials Actual $59,400...
-
For the house data in Table 4.1, the average selling price is y = 447.0. Imagine that you do not know the size of any house, so you predict the selling price of each of them to be 447.0. This is...
-
2.) Find the Laplace transform of f(t) 7e-St cos 2t +9 sinh2 2t. Use Laplace Table. %3D
-
Show that the average velocity of a car over a time interval [t 1 , t 2 ] is the same as the average of its velocities during the trip.
-
TMS ethers can be removed by treatment with fluoride ion as well as be acid-catalyzed hydrolysis. Propose a mechanism for the reaction of cyclohexyl TMS ether with LiF. Fluorotrimethyisilane is a...
-
Show the mechanism of the reaction of p-methaylphenol with 2-methylpropene and H3PO4 catalyst to yield the food additive BHT.
-
Assume that you need to prepare 5-cholesten-3-one from cholesterol. How could you use IR spectroscopy to tell whether the reaction was successful? What differences would you look for in the IR...
-
During December, Willow Appliance Company sold appliances to Ragnar Company for $25,000. Willow is unable to determine Ragnar's ability to pay the amount owing. Ragnar pays the full amount due in...
-
content area top Part 1 On March 1, 2024, Barker Services issued a 2% longminusterm notes payable for $ 20 comma 000. It is payable over a 5minusyear term in $4 comma 000 annual principal payments on...
-
Of what use is probability determination in (1) card playing, (2) other gaming or gambling, or (3) lottery draws? Are deciding with the odds worth the risk to a certain point? Are deciding against...

Study smarter with the SolutionInn App


