Compound A, C 4 H 8 O 3 , has infrared absorptions at 1710 and 2500 to
Question:
Compound A, C4H8O3, has infrared absorptions at 1710 and 2500 to 3100 cm?1 and has the 1H NMR spectrum shown. Propose a structure for A.
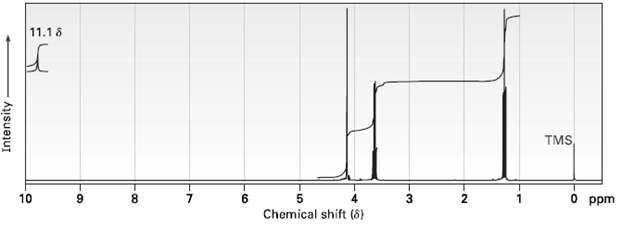
Transcribed Image Text:
11.18 TMS 10 O ppm 8. 6. 3. Chemical shift (6) Intensity -00
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 90% (11 reviews)
The compound has one degree of ...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The 1H NMR spectrum shown is that of an ether with the formula C4H8O. Propose astructure. TMS 10 9. 8. 7. 6. O ppm Chemical shift (8) Intensity 3.
-
The 1H NMR spectrum shown is that of 3-methyl-3-buten-l-ol. Assign all the observed resonance peaks to specific protons, and account for the splittingpatterns. TMS H H2H20 10 8. O ppm Chemical shift...
-
The 1H NMR spectrum of compound A (C8H8O) consists of two singlets of equal area at 5.1 (sharp) and 7.2 ppm (broad). On treatment with excess hydrogen bromide, compound A is converted to a single...
-
A 100-m-long conductor of uniform cross section has a voltage drop of 4 V between its ends. If the density of the current flowing through it is 1.4 x 106 (A/m2), identify the material of the...
-
1. As a strategic leader, what error might Mary Briggs be making? 2. To what extent should a school superintendent be spending more time on strategy than operational problems? 3. What advice might...
-
Suppose the number of baskets scored by the Indiana University basketball team in one minute follows a Poisson distribution with = 1.5. In a 10-minute span of time, what is the probability that...
-
On December 1, 2020, Junction Company issued at 104, 4,000 of its \(9 \%, 10\)-year, \(\$ 1,000\) par value, nonconvertible bonds with detachable stock purchase warrants. Each bond carried two...
-
As of June 30, 2016, the bank statement showed an ending balance of $19,500. The unadjusted Cash account balance was $15,200. The following information is available: 1. Deposit in transit, $2,400. 2....
-
A real estate investor feels that the cash flow from a propertywill enable her to pay a lender $15,000 per year, at the end ofevery year, for 10 years. How much should the lender be willing toloan 2...
-
For each of the following independent scenarios, determine whether Van Allen Corporation is a principal or an agent for purposes of applying the 5-step revenue recognition model. 1. Van Allen is a...
-
How would you use NMR (either 13C or 1H) to distinguish between the following pairs of isomers? (a) C and 2 (b) 2CH2CH2CO2H CH3CH(CO2H)2 and (c) CH3CH2CH2C02H and H-CH2CH2CH and -2 (d)...
-
Propose a structure for a compound C4H7N that has the following IR and 1H NMR spectra: 100 80 - 60 40 - 20 - 4000 3500 3000 2500 2000 1500 1000 500 Wavenumber (cm-1) TMS O ppm 10 9. 8. 3. Chemical...
-
Compute the median and mean for Distribution 6 in Table 3.2. Why can't you find the mode of this distribution? Table 3.2 Distribution 3 6. 4 7.0 7.2 40.2 7.0 10.0 97.8 7.0 7.1 40.1 7.0 10.0 88.5 7.0...
-
How do clearly provides a personal WHY STATEMENT reflecting your personal brand as a business management professional on Linked - clearly shows how you have demonstrated a GROWTH MINDSET with...
-
Charges 1-4 are placed one at a time in the presence of a potential due to a larger positive charge. Rank the electric potential energy due to the interaction of charges 1-4 individually with the...
-
Introduction: In your introduction, include why you chose Dream Diaper by Bambo Nature for this paper. What is the need? How does this product fill a market need? What is innovative about it? How is...
-
A human resource department is evaluating its pay structure. A compensation specialist computes the compa-ratio of the designers and determines that it is 1. 4. What problem is most likely to result...
-
Marketing communications industry trends, including impact of new/alternative digital technologies - Discuss key trends in marketing communications. - Identify at least five and briefly describe each...
-
To compute a binomial probability, we must know both the success probability and the number n of ___________________ . In Exercises 57, fill in each blank with the appropriate word or phrase.
-
The baseball player A hits the ball from a height of 3.36 ft with an initial velocity of 34.8 ft/s. 0.14 seconds after the ball is hit, player B who is standing 15 ft away from home plate begins to...
-
Platinum crystallizes with the face-centered cubic unit cell. The radius of a platinum atom is 139 pm. Calculate the edge length of the unit cell and the density of platinum in g/cm 3 .
-
For which of the following ions does the formal charge give a fairly accurate picture of where the charge really is? Explain in each case. (a) NH4 (b) H3O: (C) NH2 (d) CH3
-
Is there an unbranched alkane containing 23 hydrogen atoms? If so, give its structural formula; if not, explain why not.
-
In the structure of 4- isopropy 1-2,4,5-trimethylheptane (Problem 2.9) (a) Identify the primary, secondary, tertiary, and quaternary carbons. (b) Identify the primary, secondary, and tertiary...
-
Chapter 4: Estimating Demand 127 Questions 1. Estimate the demand for soft drinks using a multiple regression program available on your computer. 2. Interpret the coefficients and calculate the price...
-
[Chapter 5 - Pictorial Drawings] SP5-1) Draw a full scale isometric pictorial and a cabinet oblique pictorial by hand or in a drawing package. The answer to this problem is on the Independent...
-
Heat diffusion on a rod over the time In class we learned analytical solution of 1-D heat equation Below is the MATLAB code which simulates finite difference method to solve the above 1-D heat...

Study smarter with the SolutionInn App


