Compound A, C 8 H 10 O 2 , has an intense JR absorption at 1750 cm
Question:
Compound A, C8H10O2, has an intense JR absorption at 1750 cm?1 and gives the 13C NMR spectrum shown. Propose a structure for A.
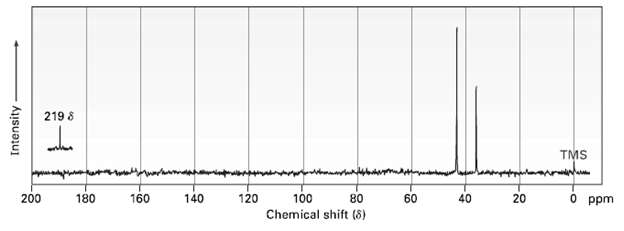
Transcribed Image Text:
219 8 TMS 200 180 160 140 120 40 O ppm 80 60 20 100 Chemical shift (8) Intensity
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (16 reviews)
Compound A has 4 degrees of unsat...View the full answer

Answered By

Akash Goel
I am in the teaching field since 2008 when i was enrolled myself in chartered accountants course
Since then i have an experience of teaching of class XI, XII, BCOM, MCOM, MBA, CA CPT.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Does the 13C NMR spectrum shown in Figure 22.10 correspond to that of 1-amino-2 methyl- 2-propanol or to 2-amino-2-methyl-1-propanol? Could this compound be prepared by reaction of an epoxide with...
-
Does the 13C NMR spectrum shown in Figure 22.10 correspond to that of 1-amino-2-methyl-2-propanol or to 2-amino-2-methyl-1-propanol? Could this compound be prepared by reaction of an epoxide with...
-
Assign the resonances in the 13C NMR spectrum of methyl propanoate, CH3CH2CO2CH3(figure). TMS CH-cH 2 1 120 200 180 160 140 100 20 40 O ppm 60 Chemical shift (8) Intensity
-
SAE 10 oil at 20C flows in a vertical pipe of diameter 2.5 cm. It is found that the pressure is constant throughout the fluid. What is the oil flow rate in m3/h? Is the flow up or down?
-
Why is SAS rated as a top company to work for by Forbes? How does SAS attract and keep top talent?
-
The federal government awarded grants to the agricultural departments of 9 universities to test the yield capabilities of two new varieties of wheat. Each variety was planted on a plot of equal area...
-
If the rate of inflation is expected to increase, would this increase or decrease the slope of the yield curve? AppendixLO1
-
Why are there two stages of allocation in activity-based costing?
-
You need $12,000 to purchase a used car. Your wealthy uncle is willing to lend you the money as an amortized loan. He would like you to make annual payments for 5 years, with the first payment to be...
-
Steller Systems completed the following stock issuance transactions: May 19 Issued 1,700 shares of $3 par value common stock for cash of $10.50 per share. Jun. 3 Issued 300 shares of $9, no-par...
-
Propose structures for molecules that meet the following descriptions. Assume that the kinds of carbons (1, 2, 3, or 4) have been assigned by DEPT-NMR. (a) C 6 H 12 O; IR: 1715 cm 1 ; 13 C NMR: 8.0 ...
-
Propose structures for ketones or aldehydes that have the following 1 H NMR spectra: (a) C 4 H 7 C1O ??IR: 1715 cm ?1 ? (b) C 7 H 14 O ? ? ?IR: 1710 cm ?1 ? (c) C 9 H 10 O 2 ? ? ?IR: 1695 cm ?1 ?...
-
Johnson & Johnson is one of the worlds largest manufacturers of health care products. The companys July 2, 2017, financial statements included the following information in the long-term debt...
-
Question (4) seen, 20 vehicles/km moving at 100 km/h and 30 vehicles/km traveling at 120 km/h. Two successive videos showing stationary traffic on the road were examined. Two groups of platoons were...
-
?In civil engineering, what is the main use of a slump test in concrete technology?
-
Explain the process of compression resin transfer molding(CRTM)?in composite manufacturing. What are the benefits of using CRTM for producing composite structures?
-
Explore the role of post-occupancy evaluation in commercial and industrial architecture. How do architects use feedback from building users to improve future designs?
-
Discuss the principles of geotechnical engineering in slope stability analysis. How can engineers assess slope stability, mitigate landslide risks, and design effective stabilization measures to...
-
Select an organization with which youre familiar either as an employee or perhaps as a frequent customer. Assume youre the top manager in this organization. What types of information from...
-
What is your opinion of advertising awards, such as the Cannes Lions, that are based solely on creativity? If you were a marketer looking for an agency, would you take these creative awards into...
-
The molecule H 2 O 2 has a resultant dipole moment of 2.2 D. Can this molecule be linear? If not, describe a shape that might account for this dipole moment.
-
Show how you might prepare each of the following amines through reductive amination: (a) (b) (c) NH2 CH3 CH3
-
When phenyl isothiocyanate, C6H5N == C == S, is reduced with lithium aluminum hydride, the product formed has these spectral data: MS (m/z): 107, 106 IR (cm-1): 3330 (sharp), 3050, 2815, 760, 700 1H...
-
When N,N'-diphenylurea (A) is reacted with tosyl chloride in pyridine, it yields product B. The spectral data for B include: MS (m/z): 194 (M+.) IR (cm-1): 3060, 2130, 1590, 1490, 760, 700 1H NMR...
-
Green Lawn Company sells garden supplies. Management is planning its cash needs for the second quarter. The following information has been assembled to assist in preparing a cash budget for the...
-
eBook Question Content Area Comparison of Methods of Allocation Duweynie Pottery, Inc., is divided into two operating divisions: Pottery and Retail. The company allocates Power and General Factory...
-
TYBALT CONSTRUCTION Income Statement For Year Ended December 31 TYBALT CONSTRUCTION Income Statement For Year Ended December 31

Study smarter with the SolutionInn App


