Electrophilic substitution on 3-phenylpropanenitrile occurs at the ortho and Para positions, but reaction with 3-phenylpropenenitrile occurs at
Question:
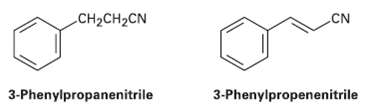
Electrophilic substitution on 3-phenylpropanenitrile occurs at the ortho and Para positions, but reaction with 3-phenylpropenenitrile occurs at the meta position. Explain using resonance structures of theintermediates.
Transcribed Image Text:
CH2CH2CN .CN 3-Phenylpropenenitrile 3-Phenylpropanenitrile
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
When directly bonded to a ring theCN group is a metadirecting dea...View the full answer

Answered By

Shaira grace
I have experience of more than ten years in handing academic tasks and assisting students to handle academic challenges. My level of education and expertise allows me communicate eloquently with clients and therefore understanding their nature and solving it successfully.
5.00+
4+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Electrophilic aromatic substitution usually occurs at the 1-position of naphthalene, also called the position. Predict the major products of the reactions of naphthalene with the following reagents....
-
The pair of structures that are resonance hybrids is (a) (b) (c) (d) HO-CHCH, and Ho=CHCH, CH2 CH and CH CH2 :O CH and CH, CH3CH2 and CH2CH
-
The following two drawings are resonance structures of one compound: But the following two drawings are not resonance structures: They are, in fact, two different compounds. Explain. Not resonance...
-
Did you have imaginary friends when you were a young child? What do you remember about them and their value to you?
-
Gus, who is married and files a joint return, owns a grocery store. In 2016, his gross sales were $276,000, and operating expenses were $320,000. Other items on his 2016 return were as follows:...
-
The file Bulbs contains the life (in hours) of a sample of 40 100-watt light bulbs produced by Manufacturer A and a sample of 40 100-watt light bulbs produced by Manufacturer B. The following table...
-
Jerome and Gerard, Certified Public Accountanis, are preparing for the annual audit of the Fordham Office Machine Company. Several procedures which are included in the audit program pertaining to...
-
Use the Sudan Medical Supply Corp. data from P12- 62A. The 2014 and 2013 comparative balance sheets and 2014 income statement of Sudan Medical Supply Corp. Follow: Requirements 1. Prepare the 2014...
-
Laura is the controller for Wildhorse Inc., a factory that creates unique dog chew toys. Until now, Wildhorse has been using a single plant-wide rate of $10.60 per direct labor hour to allocate...
-
A group of your friends and you are very excited about setting up a business, in Indonesia selling imported home accessories and furniture sourced from all over the world. There are eight of you, who...
-
At what position and on what ring do you expect nitration of 4-bromo- biphenyl to occur? Explain, using resonance structures of the potentialintermediates. 4-Bromobiphenyl Br
-
Addition of HBr to 1-phenyipropene yields only (1-bromopropyl) benzene. Propose a mechanism for the reaction, and explain why none of the other regioisomer isproduced. Br + HBr
-
1. Why did Young choose to solve the autocorrelation problem first? 2. Would it have been better to eliminate multicollinearity first and then tackle autocorrelation? 3. How does the small sample...
-
Let \ theta be the angle that the vector A - > makes with the + x + - axis, measured counterclockwise from that axis. Find the angle \ theta for a vector that has the following components. Ax = 4 . 1...
-
The mass of a large concrete block is 5 5 8 kg . Is this a scalar quanity? explain
-
Imagine you are traveling in a wide open space. You walk 2 0 . 0 m due north, and then 1 7 . 0 m due east. what is your displacement start to finish?
-
How would 3PLs score on these criteria against competition (contract manufacturers, consultants, IT vendors, transport companies and freight forwarders)? Or in other words: what are the critical...
-
Assume that The AM Bakery is preparing a budget for the month ending October 31. Management prepares the budget by starting with the actual results for August 31. Next, management considers what the...
-
Obtain from a licensed salesperson or stockbroker a prospectus and any other literature available on a popular deferred annuity. Analyze the terms, and answer the following questions. a. What is the...
-
A bubble-point liquid feed is to be distilled as shown in Figure. Use the Edmister group method to estimate the mole-fraction compositions of the distillate and bottoms. Assume initial overhead and...
-
For real gases the Joule-Thomson coefficient is greater than zero at low temperatures and less than zero at high temperatures. The temperature at which is equal to zero at a given pressure is called...
-
Show the products of thesereactions: Bra Br2 a) CH,CH,CH=CH2 b) CH Cl2 CH Cl2 excess Cl2 CH-Cl2 Brz CCI4 d)
-
Show all of the steps, including stereochemistry, in the mechanism for thisreaction: Br + Br2 Br
-
Explain which of these compounds has the faster rate of reaction withBr2: Ph Ph - c=CH, or .
-
A 2-meter-tall basketball player attempts a goal 12.4 meters from the basket that is 3.05 meters high. If he shoots the ball at a 55 angle, at what initial speed must he throw the basketball so that...
-
Find the following. 2. 3 y(t) + 3y(t) 3+ = x(t) + 3 x(t) 3. y(t) - 1/2 y(t) x(t) = dt d 4. dt d 5. dt y(t) + 1/2 y(t) + y(t) y(t) + 1/2 y(t) = x(t) + x(t) = x(t)
-
1. Who are the people affected by rsum truth decisions? 2. Why might a job seeker have a duty to blur parts of his or her work history? 3. Why might an egoist lie on the rsum, and why not? 4. Does an...

Study smarter with the SolutionInn App


