How would you carry out the following transformations? Tell the reagents you would use in each case.
Question:
How would you carry out the following transformations? Tell the reagents you would use in each case.
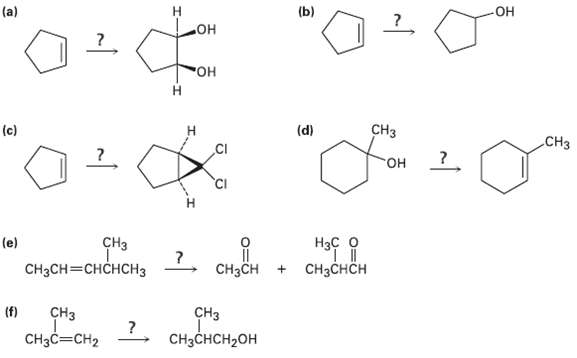
Transcribed Image Text:
(a) (b) Он но° "Он н CHз (c) (d) CHз но- CHз CH3CH=CHCHCH3 (e) Нас о ?. CнзCH + CHзснсH (f) CHз CHз CHзсHCH-оH CH3C=CH2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 91% (12 reviews)
a b OsO4 NMO H I OH OH OH 1 HgOAc2 H2O 2 NaBH4 Hy...View the full answer

Answered By

Brown Arianne
Detail-oriented professional tutor with a solid 10 years of experience instilling confidence in high school and college students. Dedicated to empowering all students with constructive feedback and practical test-taking strategies. Effective educator and team player whether working in a school, university, or private provider setting. Active listener committed to helping students overcome academic challenges to reach personal goals.
4.60+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How would you carry out the following transformations using an organo copper coupling reaction? More than one step is required in eachcase. (a) "CH (b) HH2CH2CHBr CH3CH2CH2CH2CH2CH2CH2CH3 (c)...
-
How would you carry out the following transformations? More than one step may berequired. H-CH (a) (b) Br - (c) " - " - (d) CH3CH2CH2CH2C=CH CHH2CH2CH2CH2H20CH3 H (e) CH3CH2CH2CH2CHCH3...
-
How would you carry out the following transformation, a step used in the commercial synthesis of(S)-ibuprofen? CN
-
Show that the angular wave number k for a non-relativistic free particle of mass m can be written as in which K is the particle's kineticenergy 27 V2mK k =
-
Calculate each Poisson probability: a. P(X = 6), = 4.0 b. P(X = 10), = 12.0 c. P(X = 4), = 7.0
-
Compute the minimum distance of the code C and decode the vectors u, v, and w using nearest neighbor decoding. C = u =|0 |, v =| 0 w = | 0
-
14. When an employer provides group-term life insurance to an employee, what are the tax consequences to the employee? What are the tax consequences to the employer?
-
1. To what extent were the problems facing the notebook computer development project avoidable? What could have been done to avoid these problems? 2. Would it make sense to apply a project selection...
-
Gladstorm Enterprises sells a product for $53 per unit. The variable cost is $35 per unit, while fixed costs are $19,836. a. Determine the break-even point in sales units. Round answer to the nearest...
-
XYZ Corp sells widgets to consumers for $20 each. Its beginning accounts receivable balance was $24,975, and it sold 12,376 widgets throughout the year. The total cash collections for the year...
-
Predict the products of the following reactions, showing both regiochemistry and stereochemistry where appropriate: CH (b) (a) 1. 2. Zn, H* M H* CH 1. Hg(OAc)2, H20 2. NABH4 (c) (d) C 1. 2. Hz, "
-
Which reaction would you expect to be faster, addition of HBr to cyclohexene or to 1-methylcyclohexane? Explain.
-
A five-year credit default swap requires quarterly payments at the rate of 60 basis points per year. The principal is $300 million and the credit default swap is settled in cash. A default occurs...
-
A storeroom is used to organize items stored in it on N shelves. Shelves are numbered from 0 to N-1. The K-th shelf is dedicated to items of only one type, denoted by a positive integer A[K]....
-
CASES CASE 10.1 Money in Motion Jake Nguyen runs a nervous hand through his once finely combed hair. He loosens his once perfectly knotted silk tie. And he rubs his sweaty hands across his once...
-
(3.8) Axiom, Definition of false false = true (3.9) Axiom, Distributivity of over : (pq) p=q
-
The board of directors of Unilever has been impressed by the presentation you did, and they further instructed you to conduct a more insightful investigation about the Sri Lankan market. They have...
-
The sample space listing the eight simple events that are possible when a couple has three children is {bbb, bbg, bgb, ogg, gbb, gbg, ggb, ggg}. After identifying the sample space for a couple having...
-
On the Essence of Cost Control. : Cost control activities can eliminate only the continuation of out-of-line performance. This is true since no one can alter or undo what has already occurred. With...
-
Data 9.2 on page 540 introduces the dataset Cereal, which includes information on the number of grams of fiber in a serving for 30 different breakfast cereals. The cereals come from three different...
-
Methanol has a normal boiling point of 64.6 C and a heat of vaporization (H vap ) of 35.2 kJ/mol. What is the vapor pressure of methanol at 12.0 C? SORT You are given the normal boiling point of...
-
Predict the products, including their stereochemistry, from the E2 reactions of the following diastereomers of stilbene dibromide with sodium ethoxide in ethanol. Assume that one equivalent of HBr is...
-
Draw the structure of the starting material that would undergo azir-elimination give the -E isomer of the alkene product in the E2 reaction of Eq. 9.40.
-
Draw the structure of the starting material that would undergo azir-elimination give the -E isomer of the alkene product in the E2 reaction of Eq. 9.40.
-
Portfolio return and beta Personal Finance Problem Jamie Peters invested $ 1 1 3 , 0 0 0 to set up the following portfolio one year ago: a . Calculate the portfolio beta on the basis of the original...
-
. Emerson Cammack wishes to purchase an annuity contract that will pay him $7,000 a year for the rest of his life. The Philo Life Insurance Company figures that his life expectancy is 20 years, based...
-
Integrity Inc. can sell 20-year, $1,000 par value bonds paying semi-annual interests with a 10% coupon. The bonds can be sold for $1,050 each; flotation cost of $50 per bond will be incurred in this...

Study smarter with the SolutionInn App


