Hydrocarbon A, C 10 H 14 , has a UV absorption at ? max = 236 nm
Question:
Hydrocarbon A, C10H14, has a UV absorption at ? max = 236 nm and gives hydrocarbon B, C10H18, on catalytic hydrogenation. Ozonolysis of A followed by zinc/acetic acid treatment yields the following diketo dialdehyde:
(a) Propose two possible structures for A.
(b) Hydrocarbon A reacts with maleic anhydride to yield a Diels?Alder adduct. Which of your structures for A is correct?
(c) Write the reactions, showing starting material and products.
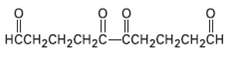
Transcribed Image Text:
ененонй оеко onрнм нссH-CH2CH2C —ҫсн-CH2CH2CH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (7 reviews)
a Hydrocarbon A must have two double bonds and two rings since the sole o...View the full answer

Answered By

Brown Arianne
Detail-oriented professional tutor with a solid 10 years of experience instilling confidence in high school and college students. Dedicated to empowering all students with constructive feedback and practical test-taking strategies. Effective educator and team player whether working in a school, university, or private provider setting. Active listener committed to helping students overcome academic challenges to reach personal goals.
4.60+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Propose two possible structures for a compound with molecular formula C 5 H 12 O that exhibits the following 13 C NMR and IR spectra: Carton 13 NMR 29.1 9.5- 73.8- 100 90 80 70 60 50 20 10 Chemical...
-
Propose two possible structures for a compound with molecular formula C 5 H 8 that produces an IR signal at 3300 cm -1 .
-
The following triene reacts with excess maleic anhydride to produce a compound with molecular formula C 14 H 12 O 6 . Draw the structure of this product (ignoring stereochemistry). Maleic anhydride...
-
Lamonda Corp. uses a job order cost system. On April 1, the accounts had the following balances: The following transactions occurred during April: (a) Purchased materials on account at a cost of...
-
How do you predict these pay rate changes at Parkland Health will affect its performance?
-
List and describe study skills employees should learn before beginning a training program.
-
When should an organization consider conducting independent audits?
-
The defendant agreed to sell watches to the buyer in Mexico. A notation was printed at the bottom of the contract, which, translated into English, reads as follows: Please send the merchandise in...
-
A 15 year, 8% coupon rate bond is currently trading at $958. The yield to maturity of this bond must be greater than 8% equal to 8% less than 8% unknown
-
It is October 16, 2020, and you have just taken over the accounting work of China Moon Products, whose annual accounting period ends October 31. The company?s previous accountant journalized its...
-
Addition of HC1 to 1-methoxycyclohexene yields 1-chloro-1-methoxycyclo- hexane as the sole product. Use resonance structures to explain why none of the other regioisomer isformed. HCI CI CI
-
Adiponitrile, a starting material used in the manufacture of nylon, can be prepared in three steps from 1, 3-butadiene. How would you carry out thissynthesis? 3 steps H2C=CHCH=CH2 N=CCH2CH2CH2CH2C=N...
-
Why does register renaming prevent write-after write hazards (WA W) but not read-after-write (RAW) hazards?
-
1. 1.1. 1.2. 1.3. The U.S. small motor boat industry is clustered in California. The boats are sold in the competitive world market, at a world price of $5,000 per boat. The annual supply curve for...
-
where x=your a) Consider a scenario in which you are given an IP address in the following format: 'x.0.0.0', roll no. + 110. (E.g., if your roll no. is 35, then x=35+110=145, and the IP address will...
-
Discuss in detail and be sure to discuss the important points. What is a bond? Describe the various types of risks for bonds. How do you value a Bond? Provide a numerical example of how to value a...
-
For the month of November, AC3220 Ltd. recorded $500,000 in sales, 40% of which were on account (terms N30), and 60% of which were cash sales. The company is required to charge 15% HST on all sales....
-
The boardof directors ofAPI, a relatively new electronicsmanufacturer, has decided to continue paying a common stock dividend to increase the attractiveness of the stock in the free market. The board...
-
Firm F manufactures and sells two products: A and B. (Exercise courtesy of Prof. Mara Jess Grandes from IESE Business School.) Parameters for both products are as follows: Note that fixed costs are...
-
The age-old saying for investing is "buy low and sell high," but this is easier said than done. Investors who panic about falling prices sell their investments, which in turn lowers the price and...
-
What is the molarity of a 6.56% by mass glucose (C 6 H 12 O 6 ) solution? (The density of the solution is 1.03 g/mL.) SORT You are given the con- centration of a glucose solution in percent by mass...
-
(a) Simple aminoacetals hydrolyze quickly and easily in dilute acid. Propose a mechanism for hydrolysis of the following aminoacetal: (b) The nucleosides that make up DNA have heterocyclic rings...
-
The mass spectrum of unknown compound A shows a molecular ion at m/z 116 and prominent peaks at m/z 87 and m/z 101. Its UV spectrum shows no maximum above 200 nm. The IR and NMR spectra of A follow....
-
Show how you would accomplish the following synthetic conversions by adding an organolithium reagent to an acid. (a) (b) (c) Pentanoic acid heptan-3-one (d) Phenylacetic acid...
-
What does it mean to be fully vested in a pension or 4 0 1 k retirement plan?
-
Which items should be considered as cost of goods sold, initial expenditure, EBITDA, depreciation, EBIT, interest, fixed assets, and total current assets for daycares?
-
R&R construction Limited supplies for April 2024 was: Construction Services $500,000 ( exempt VAT activity) Electrical Services $2,000,000 Surveying Services $300,000 Export Services $700,000 The VAT...

Study smarter with the SolutionInn App


