a. Draw the displayed formulae and name the functional group isomers of C 3 H 6 O
Question:
a. Draw the displayed formulae and name the functional group isomers of C3H6O that are:
i. An aldehyde
ii. A ketone.
b. Draw the displayed formula and name an isomer of C3H8O that could be used as an example of positional isomerism of one of the isomers in Figure 14.17.
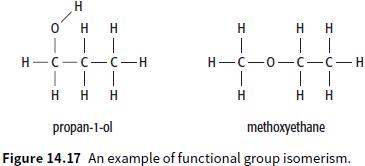
Transcribed Image Text:
H o' H H H H H 1. Н—С — о—с—с — н Н—с — с—с —н H H H H H H propan-1-ol methoxyethane Figure 14.17 An example of functional group isomerism.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 42% (7 reviews)
1 The displayed formula for C3H60 which contains ketone and aldehyde ...View the full answer

Answered By

Samee Ullah
Algebra, Linear algebra, calculus, accounting, marketing, statistics, programming, real estate, writing, human resource management, business communication, Engineering: civil, chemical, electrical, mechanical, aerospace, building
Linguistics: sociolinguistics, applied linguistics, music, social sciences, biology, chemistry: all types, Thermodynamics, mechanics, modern physics, quantum physics, metaphysics, biology.
Feel free to contact us for all these subjects,; for quality, and best responses. Thankyou
0.00
0 Reviews
10+ Question Solved
Related Book For 

Cambridge International AS And A Level Chemistry Coursebook
ISBN: 9781316637739
2nd Edition
Authors: Lawrie Ryan, Roger Norris
Question Posted:
Students also viewed these Sciences questions
-
a. Name the four isomers in Figure 14.15. b. Draw the displayed formulae and name the structural isomers of C 3 H 7 Cl. Br Br H Br H Br H-C-C-C-H H H H H H H Br H H Br H-C-c-c-H Br H H H Br H...
-
a. Draw the displayed formulae and name the cistrans isomers of but-2-ene. b. The molecule CHBrClF exhibits optical isomerism. Draw the 3D displayed formulae of both optical isomers.
-
Draw the displayed formulae and give the names of the isomers of C 5 H 12 .
-
Air flows with negligible friction through a 4-indiameter duct at a rate of = lbm/s. The temperature and pressure at the inlet are T1 = 800 R and P1 = 30 psia, and the Mach number at the exit is Ma2...
-
A random sample of 49 measurements from one population had a sample mean of 10, with sample standard deviation 3. An independent random sample of 64 measurements from a second population had a sample...
-
A study analyzed the behavior of the stock prices of firms that had lost antitrust cases. Included in the diagram are all firms that lost the initial court decision, even if the decision was later...
-
Silicon Solutions Inc. manufactures RAM memory chips for personal computers. An activity analysis was conducted, and the following activity costs were identified with the manufacture and sale of...
-
Beckett Corporation realized $800,000 of taxable income from the sales of its products in States A and B. Beckett's activities establish nexus for income tax purposes in both states. Beckett's sales,...
-
Needing to record the following merchandise inventory transactions, ACME Limited wants your help with recording all purchase/sales discounts for early payment when they make their payments. Ignore...
-
Stream cross-sectional areas ( A ) are required for a number of tasks in water resources engineering, including flood forecasting and reservoir designing. Unless electronic sounding devices are...
-
Draw a 3D formula for: a. Propane b. Propene.
-
a. Write an equation to show the homolytic fission of the Cl Cl bond in a chlorine molecule, Cl 2 . b. Write an equation to show the heterolytic fission of the C Cl bond in chloromethane. Include a...
-
May Manufacturing Company was organized January 2, 2007. During 2007, it has used in its reports to management the straight-line method of depreciating its plant assets. On November 9, you are having...
-
Question: 9. Purchases and sales during a recent period for Bottineau Inc. were Purchases During the Period Sales During the Period 1st purchase 1,500 units x $ 4 1st sale 700 units x $13 2nd...
-
# The following is a partial relative frequency distribution of consumer preferences for four products-A, B, C, and D. Required: Determine the relative frequency for Product B: Relative Frequency...
-
Domino Company's operating percentages were as follows: Revenues 100% Cost of goods sold Variable 50% Fixed 10% 60% Gross profit 40% Other operating expenses Variable 20% Fixed 15% 35% Operating...
-
Marcus Stewart, the production manager at Galvin Company, purchased a cutting machine for the company last year. Six months after the purchase of the cutting machine, Stewart learned about a new...
-
The TechTeach Company produces and sells 7,000 modular computer desks per year at a selling price of $750 each. Its current production equipment, purchased for $1,950,000 and with a 5-year useful...
-
On January 1, 2021, Cameron Inc. bought 20% of the outstanding common stock of Lake Construction Company for $300 million cash, giving Cameron the ability to exercise significant influence over Lakes...
-
(a) What is the focal length of a magnifying glass that gives an angular magnification of 8.0 when the image is at infinity? (b) How far must the object be from the lens?
-
When 2-bromo-2-methylhexane is treated with sodium ethoxide in ethanol, the major product is 2-methyl-2-hexene. a) Draw the mechanism of this reaction. b) What is the rate equation of this reaction?...
-
Predict the major product for each of the following reactions: a. b. c. d. e. f. g. h. i. j. CI :? NaSH DBN OTs
-
Draw all constitutional isomers of C 4 H 9 Br, and then arrange them in order of increasing reactivity toward an E2 reaction.
-
Required : a- outline the statement of comperhensive income for the year ended 30 november 2021 b- outline the statment of financial position as at 30 November The Trial Balance of Alim Enterprise at...
-
International business and environment The MIR requires teams to gather current, or the most recently available, data on the markets people, economy, government, and technological status from online...
-
Consider the following stream of cash flows. The interest rate is 10%. 0 1 2 3 4 5 6 7 100 100 100 200 0 300 300 300 a) What is the value at time 0 of the cash flow stream? b) What is the value of...

Study smarter with the SolutionInn App


