Repeat Problem 5.9 assuming that helium is described by the Peng-Robinson equation of state. Problem 5.9 High-pressure
Question:
Repeat Problem 5.9 assuming that helium is described by the Peng-Robinson equation of state.
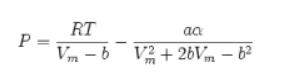
Problem 5.9
High-pressure helium is available from gas producers in 0.045-m3 cylinders at 400 bar and 298 K. Calculate the explosion equivalent of a tank of compressed helium in terms of kilograms of TNT. Assume helium is an ideal gas.
Transcribed Image Text:
P = RT aa Vm-b V2+2bVm – b²
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
To solve this problem we will need to use the PengRobinson equation of state to calculate the fugacity of helium at the given conditions The fugacity ...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Chemical Biochemical And Engineering Thermodynamics
ISBN: 9780470504796
5th Edition
Authors: Stanley I. Sandler
Question Posted:
Students also viewed these Engineering questions
-
High-pressure helium is available from gas producers in 0.045-m 3 cylinders at 400 bar and 298 K. Calculate the explosion equivalent of a tank of compressed helium in terms of kilograms of TNT....
-
In this problem, we consider the analysis of the combined information from both raters on the shoulder flexion (SF) scores in the posture measurement study. Thus, the questions below concern the data...
-
The molar heat capacity C P,m of SO 2 (g) is described by the following equation over the range 300 K < T < 1700 K: In this equation, T is the absolute temperature in kelvin. The ratios T/K ensure...
-
Suppose you make beautiful coffee tables out of juniper trees. Your materials cost for each table is $135. You pay your craftsman $25 per hour and it takes him 5 hours to complete one table. If you...
-
Several manufacturers introduced into the American market products known as all-terrain vehicles (ATVs). ATVs are motorized bikes that sit on three or four low-pressure balloon tires and are meant to...
-
As a group, collect recent job advertisements from newspapers and Internet sites. Establish the necessary criteria and the desirable criteria based on one or more of the advertisements and record...
-
AutoAnswer, Inc., a manufacturer of telephone answering machines, uses a factory overhead applied rate to charge overhead costs to its manufactured products. The company manager estimates that...
-
Compute Income Measures Presented below is information related to Viel Company at December 31, 2010, the end of its first year of operations. Sales revenue...................... 310,000 Cost of goods...
-
Dan is a 16 year-old who decided to skip his adolescent development class. If Dan was 19 years-old, this would be his choice, but because of his age, he has broken the law. What type of offence did...
-
Please assume figures are USD currency unless otherwise specified and provide all answers in USD. 1 You've been given the following information: - Market/reference prices for a variety of metals and...
-
Using the Redlich-Kwong equation of state, compute and plot (on separate graphs) the pressure of nitrogen as a function of specific volume at the two temperatures: a. 110 K b. 150 K
-
The Joule-Thomson coefficient, , given by is a function of temperature. The temperature at which = 0 is known as the inversion temperature. a. Use the van der Waals equation of state to determine...
-
On February 18, 2013, Union Corporation purchased 10,000 shares of IBM common stock as a long-term investment at $60 per share. On December 31, 2013, and December 31, 2014, the market value of IBM...
-
Jessica bought a new heavy truck for $35,000 to use 80% for her sole proprietorship. Total miles driven include 8,000 in 2019, 9,000 in 2020, and 8,500 in 2021. If Jessica uses the standard mileage...
-
in January 2018 photo studios acquires new camera. The price of the cameras is 1750 how can i calculate the impairment loss of the camera at the end of 2020, taking into account that the recoverable...
-
Using the EOQ Model, Tokyo Company computed the economic order quantity for one of the materials it uses in its production to be 4,000 units. The Company maintains safety stock of 300 units. The...
-
Future Value of an Annuity What is the future value of a $1,300 annuity payment over 6 years if the interest rates are 9 percent?
-
Assume an investor is looking for financing for a project that costs $300,000. A lender offers financing available in the amount of $210,000 at an annual mortgage constant of 10.00%. The loan is...
-
The Code contains provisions that are friendly to specific groups of taxpayers. Among these are the following: Seniors. Married taxpayers. Employed taxpayers. Taxpayers with children. ...
-
What are the risks and liability factors in an audit? What are the implications to the auditor? What are the implications to the organization? How can the auditor mitigate these risks and liability...
-
Use a plot over the range 0 x 5 to con rm that sin( ix) = i sinh x.
-
The function y(t) = 1 -! e -b t, where t is time and b > 0, describes many processes, such as the height of liquid in a tank as it is being filled and the temperature of an object being heated....
-
The following functions describe the oscillations in electric circuits and the vibrations of machines and structures. Plot these functions on the same plot. Because they are similar, decide how best...
-
Your firm, ICT Outsourced Solutions Provider (ICTISP), provides outsourced ICT support and helpdesk services to all SME's across Australia. Your services were recently acquired by AIBT. In this...
-
Helena and Daniel were divorced under an agreement executed on July 1, 2023. The terms of the agreement provide that Helena will transfer to Daniel her interest in a rental house worth $300,000 with...
-
Here are some important figures from the budget of Nashville Nougats, Inc., for the second quarter of 2 0 1 8 : April May June Credit sales $ 3 2 2 , 0 0 0 $ 3 0 2 , 0 0 0 $ 3 6 2 , 0 0 0 Credit...

Study smarter with the SolutionInn App


