Dinitrogen oxide, N 2 O, colloquially called laughing gas, was first used as an anesthetic in dentistry
Question:
Dinitrogen oxide, N2O, colloquially called “laughing gas,” was first used as an anesthetic in dentistry in 1844. Suppose that you are a chemist attempting to prepare N2O from N2 and O2; you might want to know the expected equilibrium composition. You plan to transfer a mixture of 0.482 mol N2 and 0.933 mol O2 to a reaction vessel of volume 10.0 L, where it will form N2O at 800. K; at this temperature, K = 3.2 * 10–28 for the reaction 2 N2(g) + O2(g) ⇌ 2 N2O(g). Calculate the partial pressures of the gases in the equilibrium mixture.
ANTICIPATE Because the equilibrium constant is so small, you should expect that equilibrium will be reached when only a very small amount of product has formed.
PLAN Use the procedure in Toolbox 5I.1. Because data are in liters and pressures are expected in bar, use the value of R that matches these units (namely, L · bar · K–1 · mol–1).
What should you assume? Assume that all the gases are ideal and, because the value of K is so small, that the amount of product formed will be so small that the change in partial pressures of the reactants is negligible.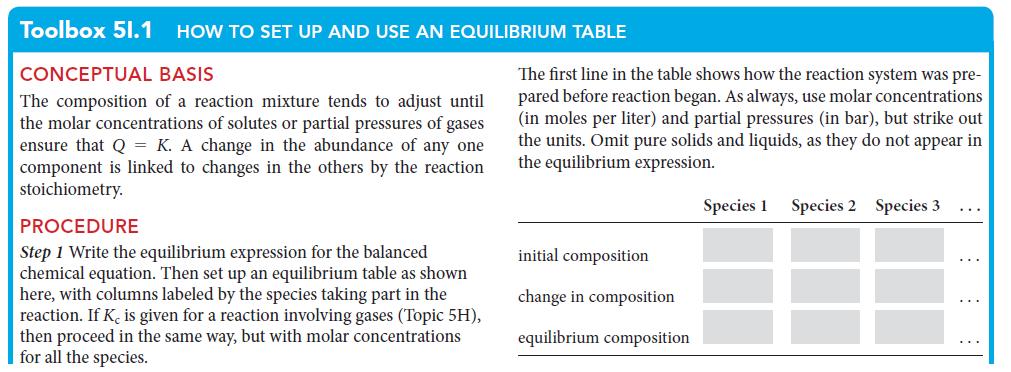
Step by Step Answer:

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman





