The values of K a for phenol and 2,4,6-trichlorophenol are 1.3 * 10 10 and 1.0 *
Question:
The values of Ka for phenol and 2,4,6-trichlorophenol are 1.3 * 10–10 and 1.0 * 10–6, respectively. Which is the stronger acid? Account for the difference in strength.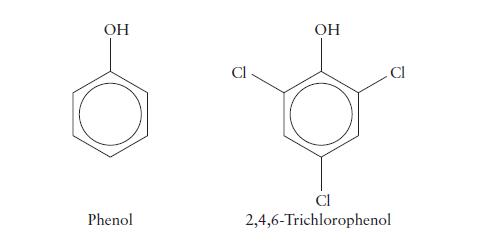
Transcribed Image Text:
OH Phenol OH Cl 2,4,6-Trichlorophenol
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
246Tric...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
As a part of your research program on formic acid, you need to titrate a solution of formic acid with sodium hydroxide solution and want to know what to expect. Calculate the pH of (a) 0.100 m...
-
The amino acid glycine (H2N-CH2-COOH) can participate in the following equilibria in water: (a) Use the values of Ka and Kb to estimate the equilibrium constant for the intramolecular proton transfer...
-
Consider the acids in Table. Which acid would be the best choice for preparing a pH = 7.00 buffer? Explain how to make 1.0 L of this buffer. Table Values of Ka for Some Common Monoprotio Acids...
-
The coordinates of points A and B are given below: Easting Northing Height (meter) (meter) (meter) 41.676 66.446 225.973 127.066 31.063 185.401 Point A B What is the bearing of AB (from A to B)?...
-
Joint products and byproducts (continuation of 16-16). Quality Chicken is computing the ending inventory values for its July 31, 2009, balance sheet. ending inventory amounts on July 31 are 15 pounds...
-
Consider the following deadlock-detection algorithm. When transaction T i , at site S 1 , requests a resource from T j , at site S 3 , a request message with timestamp n is sent. The edge (T i , T j...
-
(Appendix B) Explain the formerly used criteria that a business combination must meet to qualify as a pooling of interests. AppendixLO1
-
The upper leg length of 20- to 29-year-old males is normally distributed with a mean length of 43.7 cm and a standard deviation of 4.2 cm. (a) What is the probability that a randomly selected 20- to...
-
On January 1, Sustainable Energy Corporation issues bonds that have a $100,000 par value, mature in 8 years, and pay 12% interest per year. Interest payments are paid to bondholders semiannually on...
-
1. It is clear in the first meeting that Miguel is distracted and at some points disrespectful to Pilar in not listening to what she is saying to him. Still, Pilar does not do anything to draw him...
-
Write the chemical equations of the two proton transfer equilibria that demonstrate the amphiprotic character of (a) HCO 3 ; (b) HPO 4 2 . Identify the conjugate acidbase pairs in each case.
-
Calculate the molar solubility of silver bromide in 0.10 m KCN(aq).
-
Mean commute time in Atlanta, in minutes, using the data in CommuteAtlanta with n = 500, x = 29.11, and s = 20.72 Use StatKey or other technology to generate a bootstrap distribution of sample means...
-
Can anyone explain me how to calculate the ROI using the HISTORICAL COST NBV, the formula my instructor wants me to use is ADJ CF - HIST DEP /ASSETTOTAL - ACC DEP. And for the ROI of CURRENT COST NBV...
-
Consider the circuit to the right 3. If the total voltage supply in the circuit is 120V, and each resistor has a resistance of 400, what will the current read on each ammeter? |1= 12= 3 = 4. What...
-
1. The theory predicts the proportion of beans, in the four groups A, B, C and D should be 9:3:3:1. In an experiment among 1600 beans, the numbers in the four groups were 882, 313, 287 and 118. Does...
-
Would you recommend criminal charges in this case ( the screenshots below) and, if so, exactly which statutes against which person? Explain your reasoning (how the elements of the crime are met or...
-
check if each transaction is placed in the right place in each of the reports below and if there are any other mistakes in the different accounts after the first image which is a description of the...
-
Here is a list of life expectancies in 12 South American countries: 62,64,65,66,70,71,72,73,73,74,75,75 The mean of these data is 70, and the SD is 4.6. (You do not need to verify this.) Without...
-
For a Poisson process of rate , the Bernoulli arrival approximation assumes that in any very small interval of length , there is either 0 arrivals with probability 1- or 1 arrival with probability ....
-
Use the vapor pressures for hexane given in the following table to estimate the temperature and pressure of the triple point and also the enthalpies of fusion, vaporization, and sublimation. Phase T...
-
Show the paths n o p q and a b c d e f of the PVT phase diagram of Figure 8.15 in the PT phase diagram of Figure 8.4. Figure 8.4 Figure 8.15 Critical - point Liquid Solid Triple point Gas Tm...
-
Are the two PT phase diagrams below likely to be observed for a pure substance? If not, explain all features of the diagram that will not be observed. a. b. Liquid Solid Vapor Vapor Liquid Solid
-
Each week you must submit an annotated bibliography. Entries of current events relating to the economic concepts and the impact on the company or the industry of your company. You must use acceptable...
-
Fluffy Toys Ltd produces stuffed toys and provided you with the following information for the month ended August 2020 Opening WIP Units 5,393 units Units Started and Completed 24,731 units Closing...
-
Part A Equipment 1,035,328 is incorrect Installation 44,672 is incorrect Anything boxed in red is incorrect sents 043/1 Question 9 View Policies Show Attempt History Current Attempt in Progress...

Study smarter with the SolutionInn App


