Use the reactions and their equilibrium constants to predict the equilibrium constant for the reaction 2 A(s)
Question:
Use the reactions and their equilibrium constants to predict the equilibrium constant for the reaction 2 A(s) ⇌3 D(g).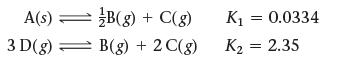
Transcribed Image Text:
A(s) = 3 D(g) B(g) + C(g) B(g) + 2 C(g) K₁ = 0.0334 K₂ = 2.35
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
To predict the equilibrium constant for the reaction 2As 3 Dg we can use ...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Consider the reactions and their respective equilibrium constants: Use these reactions and their equilibrium constants to predict the equilibrium constant for the following reaction: NO(g) + Br(g) ...
-
If CIF value is RMB 10000, calculate how much is tariff 1.Including tariff, C.T and V.A.T (1) tariff 6%, C.T 30% = ( ) (2) tariff 14%, C.T 25% = ( ) (3) tariff 70%, C.T 15% = ( ) (4) tariff 120%, C.T...
-
Carbon monoxide replaces oxygen in oxygenated hemoglobin according to the reaction: a. Use the reactions and associated equilibrium constants at body temperature given here to find the equilibrium...
-
A person who is 6 feet and 3 inches tall and weighs 185 pound force (lbf) is driving a car at a speed of 65 miles per hour over a distance of 25 miles between two cities. The outside air temperature...
-
Each of the following scenarios is independent. All cash flows are after-tax cash flows. Required: 1. Tada Corporation is considering the purchase of a computer-aided manufacturing system. The cash...
-
The figure below illustrates a thin plate of thickness \(t\). An approximate displacement field, which accounts for displacements due to the weight of the plate, is given by a. Determine the...
-
What are the basic differences between equity and debt investments, and why would equity and debt investors be interested in both earning power and solvency? Would equi ty investors tend to be more...
-
Information for 2014 follows for Rolling Thunder Corp. Rolling Thunder decided to discontinue its entire wholesale division (a major line of business) and to keep its manufacturing division. On...
-
Alpha Industries is considering a project with an initial cost of $8.2 million. The project will produce cash inflows of $1.93 million per year for 6 years. The project has the same risk as the firm....
-
Develop a database to help "YourPerfectEvent" event planners to manage their operations. The database must be able to accommodate the following. A customer contacts the company and makes...
-
Calculate K c for each reaction. a. [(g) = 21(g) Kp 6.26 x 10-22 (at 298 K) b. CH4(g) + HO(g) = CO(g) + 3 H(g) c. I(g) + Cl(g) = 2 ICI(g) = Kp = 7.7 x 1024 (at 298 K) Kp = 81.9 (at 298 K) P
-
This reaction has an equilibrium constant of Kp = 2.2 * 10 6 at 298 K. Calculate Kp for each reaction and predict whether reactants or products will be favored at equilibrium. 2 COF2(g) = CO(g) +...
-
Niedringhaus Corporations sales revenue follows: Niedringhauss management estimates that 65 percent of credit sales will be collected in the month of sale with a 2% discount taken, and that 34...
-
How do you apply what is learned in the Science of Branding 1 5 - 1 , Key Insights Regarding Global Brand Strategies Based on Research Findings to improve on the brand?
-
CASH MANAGEMENT Dr. Umburgh noticed that the $100.00 check made to Trenton Medical Supplies has not cleared for four months. What type of check is the outstanding check referred to as? a. Stale-dated...
-
Melannie Inc. sold $8,200 worth of merchandise on June 1, 2015 on credit. After inspecting the inventory, the customer determined that 10% of the items were defective and returned them to Melannie...
-
2. (20 marks) A firm wishes to produce a single product at one or more locations so that the total monthly cost is minimized subject to demand being satisfied. At each location there is a fixed...
-
Evaluate your own negotiation way. Do you have one? how you consider having an excellent negotiaiton skill could help any business person to achieve its goals.
-
Write a simulation program to price a European digital option whose underlying stock price follows a geometric Brownian motion with volatility = 0.1. Other parameters are r = 0.05, q = 0, S = K =...
-
Reread the discussion leading to the result given in (7). Does the matrix sI - A always have an inverse? Discuss.
-
A three-wire system supplies two loads A and B as shown in Fig. 2.125 . Load A consists of a motor drawing a current of 8 A, while load B is a PC drawing 2 A. Assuming 10 h/day of use for 365 days...
-
As a design engineer, you are asked to design a lighting system consisting of a 70-W power supply and two light bulbs as shown in Fig. 2.124 . You must select the two bulbs from the following three...
-
If the three bulbs of Prob. 2.59 are connected in parallel to the 120-V source, calculate the current through each bulb. Prob 2.59 An enterprising young man travels to Europe carrying three light...
-
When a loan agreement restricts a company from distributing its entire balance of retained earnings as dividends to shareholders, restricted retained earnings must be reported separately from...
-
1) A portfolio consists of 3 securities have the following characteristics in terms of standard deviation, proportion of investment and correlation coefficient. Security Standard deviation...
-
Find the future values of the ordinary annuities at the given annual rate r compounded as indicated. The payments are made to coincide with the periods of compounding. (Round your answer to the...

Study smarter with the SolutionInn App


