Use the standard reduction potentials (Appendix M) for the half-reactions [Zn(OH) 4 ] 2 (aq) + 2
Question:
Use the standard reduction potentials (Appendix M) for the half-reactions [Zn(OH)4]2−(aq) + 2 e− → Zn (s) + 4 OH−(aq) and Zn2+(aq) + 2e− → Zn(s) to calculate the value of Kformation for the complex ion [Zn(OH)4]2−.
Data given in Appendix M
Transcribed Image Text:
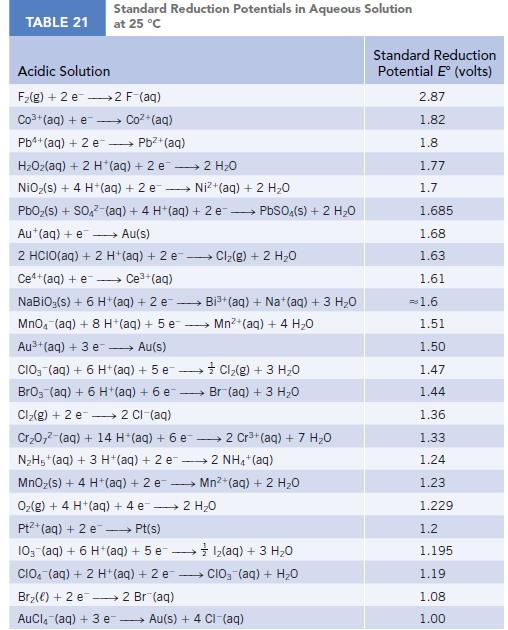
TABLE 21 Standard Reduction Potentials in Aqueous Solution at 25 °C Acidic Solution F₂(g) + 2 e 2 F-(aq) Co3+ (aq) + e Coz+(aq) Pb4+ (aq) + 2 e - Pb²+ (aq) HzOz(aq) + 2 H*(aq) +2e → 2H2O NiO₂(s) + 4 H+ (aq) + 2 e→→→→→→ Ni+(aq) + 2 HO PbO₂ (s) + SO4² (aq) + 4 H+ (aq) + 2e → PbSO4(s) + 2 H₂O Au+ (aq) + e→→→→→ Au(s) 2 HCIO(aq) + 2 H+ (aq) + 2 e-- - Ce+(aq) + e→→→→ Ce³+ (aq) NaBiO;(s) + 6 H+ (aq) + 2 e- → MnO4 (aq) + 8 H+ (aq) + 5 e Au³+ (aq) + 3 e→→→→→ Au(s) CIO3(aq) + 6 H+ (aq) + 5 e→→→→→→ BrO3 (aq) + 6 H+ (aq) + 6 e- Cl₂(g) + 2 e 2 Cl-(aq) Cr₂0,² (aq) + 14 H*(aq) + 6 e 2 Cr³+ (aq) + 7 H₂O N₂H5+ (aq) + 3 H+ (aq) + 2 e2 NH4+ (aq) MnO₂ (s) + 4 H+ (aq) + 2 e O₂(g) + 4 H+ (aq) + 4 e 2 H₂O Pt+ (aq) + 2 e →→→→→ Pt(s) 10- (aq) + 6 H+ (aq) + 5 e1₂(aq) + 3 H₂O CIO (aq) + 2 H+ (aq) + 2 e CIO₂ (aq) + H₂O Br₂() +2 e 2 Br(aq) AuCl(aq) + 3 e → Cl₂(g) + 2 H₂O → Bi³+ (aq) + Na+ (aq) + 3 H₂O Mn²+ (aq) + 4H₂O Cl₂(g) + 3 H₂O Br (aq) + 3 H₂O Mn²+ (aq) + 2 H₂O Au(s) + 4 CI-(aq) Standard Reduction Potential E (volts) 2.87 1.82 1.8 1.77 1.7 1.685 1.68 1.63 1.61 = 1.6 1.51 1.50 1.47 1.44 1.36 1.33 1.24 1.23 1.229 1.2 1.195 1.19 1.08 1.00
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)

Answered By

Hassan Imtiaz
The following are details of my Professional Experience. Responsibilities Eight years of demanding teaching experience in the field of finance and business studies at Master’s Level. Completion of the given tasks within given time with quality and efficiency. Marketing professional with practical experience in and solid understanding of a diverse range of management applications, including market analysis, sales and marketing, team building and quality assurance. I have excellent skills to approach deal and sustain corporate clients / customers by demonstrating not only extraordinary communication and interpersonal skills but also high caliber presentation, negotiation and closing skills. Manage and follow up the day-to-day activities. Manage and co-ordinate the inventories. Fulfillment of all the tasks assigned.
The following are details of my Areas of Effectiveness. Finance 1. Corporate Finance 2. Advanced Corporate Finance 3. Management of Financial Institutions 4. International Financial Management 5. Investments 6. Fixed Income 7. Real Estate Investment 8. Entrepreneurial Finance 9. Derivatives 10. Alternative Investments 11. Portfolio Management 12. Financial Statement Analysis And Reporting (US GAAP & IFRS) 13. International Financial Markets 14. Public Finance 15. Personal finance 16. Real estate 17. Financial Planning Quantitative Analysis 1. Time Value Of Money 2. Statistics 3. Probability Distribution 4. Business Statistics 5. Statistical Theory and Methods Economics 1. Principles of Economics 2. Economic Theory 3. Microeconomic Principles 4. Macroeconomic Principles 5. International Monetary Economics 6. Money and Banking 7. Financial Economics 8. Population Economics 9. Behavioral Economics International Business 1. Ethics 2. Business Ethics 3. An introduction to business studies 4. Organization & Management 5. Legal Environment of Business 6. Information Systems in Organizations 7. Operations Management 8. Global Business Policies 9. Industrial Organization 10. Business Strategy 11. Information Management and Technology 12. Company Structure and Organizational Management Accounting & Auditing 1. Financial Accounting 2. Managerial Accounting 3. Accounting for strategy implementation 4. Financial accounting 5. Introduction to bookkeeping and accounting Marketing 1. Marketing Management 2. Professional Development Strategies 3. Business Communications 4. Business planning 5. Commerce & Technology Human resource management 1. General Management 2. Conflict management 3. Leadership 4. Organizational Leadership 5. Supply Chain Management 6. Law 7. Corporate Strategy Creative Writing 1. Analytical Reading & Writing Other Expertise 1. Risk Management 2. Entrepreneurship 3. Management science 4. Organizational behavior 5. Project management 6. Financial Analysis, Research & Companies Valuation 7. And any kind of Excel Queries
4.80+
150+ Reviews
230+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Use the standard reduction potentials (Appendix M) for the half-reactions [AuCl 4 ] (aq) + 3 e Au(s) + 4 Cl (aq) and Au 3+ (aq) + 3 e Au(s) to calculate the value of K formation for the complex...
-
Use the standard reduction potentials (Appendix M) for the half-reactions Hg 2 Cl 2 (s) + 2e 2 Hg() + 2 Cl (aq) and Hg 2 2+ (aq) + 2 e 2 Hg() to calculate the value of K sp for Hg 2 Cl 2 . Data...
-
The standard reduction potentials of the following half-reactions are given in Appendix E: (a) Determine which combination of these half-cell reactions leads to the cell reaction with the largest...
-
Cummings Corp. has three business segmentsthe Plumbing Parts division, Small Tools division, and Design Printing division. Carol Jenkins (Cummings' CFO) and William Samuels (head of Production) would...
-
Gulf Coast Resins Company processes a base chemical into plastic. Standard costs and actual costs for direct materials, direct labor, and factory overhead incurred for the manufacture of 2,600 units...
-
You are on the team charged with business continuity and disaster recovery. Your role in the team is to focus on disaster recovery for the accounting system and financial data. At the first meeting,...
-
What privacy concerns might arise from the use of biometric authentication techniques? What about the embedding of RFID tags in products such as clothing?
-
Listed below are several transactions that took place during the first two years of operations for the law firm of Pete, Pete, and Roy. In addition, you learn that the company incurred utility costs...
-
Carla Vista Manufacturing Corp. decided to expand further by purchasing the net assets of Wildhorse Manufacturing Corp. Wildhorse's statement of financial position at December 31, 2023 includes the...
-
Diagram the apparatus used to electrolyze molten NaCl. Identify the anode and the cathode. Trace the movement of electrons through the external circuit and the movement of ions in the electrolysis...
-
Use standard reduction potentials (Appendix M) for the half-reactions AgBr(s) + e Ag(s) + Br (aq) and Ag + (aq) + e Ag(s) to calculate the value of K sp for AgBr. Data given in Appendix M TABLE...
-
The following is known as the transcendental production function (TPF), a generalization of the well-known CobbDouglas production function: Y i = β 1 L β2 k β3 e...
-
Saturn, Inc. is a calendar-year, accrual-basis taxpayer. The financial statements of Saturn disclose Year 4 net income of $300,000 after deducting a charitable contribution of $90,000. The...
-
Corporations need to scan the environment to evaluate opportunities and potential threats, as well as to identify strengths and weaknesses. Consider a Canadian manufacturing company that is exploring...
-
Determine the derivative for each.. a) y = (x + 1) (x +1) 3x-1 c) y = 2x+1 e) y = x-1 x+1 b) y =x(2x +3) d) y =x f) y = (4x + 1)(3+x-x).
-
1. What is the nature of the industry and the business? 2. What are the major customer groups that the Bank has chosen to focus on? 3. Determine the impact of the Bank's business strategy on its...
-
what ways does a collaborative team environment contribute to or detract from your job satisfaction?
-
Explain the expected return expression in Question 8 above.
-
Research an article from an online source, such as The Economist, Wall Street Journal, Journal of Economic Perspectives, American Journal of Agricultural Economics, or another academic journal. The...
-
A ball is thrown upward with a speed of 35 m/s from the edge of a cliff of height h - 15 m (similar to Fig. P3.22). (a) What is the speed of the ball when it passes by the cliff on its way down to...
-
Jeff Gordon (a race car driver) discovers that he can accelerate at 4.0 m/s 2 without spinning his tires, but if he tries to accelerate more rapidly, he always burns rubber. (a) Find the coefficient...
-
Anti lock brakes. A car travels at 65 mi/h when the brakes are suddenly applied. Consider how the tires of a moving car come in contact with the road. When the car goes into a skid (with wheels...
-
Given the marginal cost estimates associated with obtaining additional interest-checking account funding in the table below, what is the estimated marginal cost of obtaining additional...
-
( a ) In this problem, we will now make deposits at the beginning of the year. The only columns we will need to interchange are the interest and deposit columns. Time and ending balance formula's...
-
Adjustments for the year ending december 31 2020. climbing equipment is depreciated using the accelerated depreciation method at a rate of 20%. the only acquisition made this year was made on...

Study smarter with the SolutionInn App


