Using values from the table of standard reduction potentials, calculate the cell potentials of the following cells.
Question:
Using values from the table of standard reduction potentials, calculate the cell potentials of the following cells.
(a) ![]()
(b) 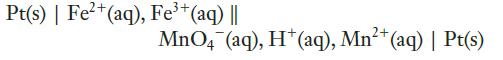
(c) ![]()
Transcribed Image Text:
Fe(s) | Fe²+ (aq) || Hg2+ (aq) | Hg()
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
To calculate the cell potentials of the given cells using the standard reduction potentials we will ...View the full answer

Answered By

Felix Onchweri
I have enough knowledge to handle different assignments and projects in the computing world. Besides, I can handle essays in different fields such as business and history. I can also handle both short and long research issues as per the requirements of the client. I believe in early delivery of orders so that the client has enough time to go through the work before submitting it. Am indeed the best option that any client that can think about.
4.50+
5+ Reviews
19+ Question Solved
Related Book For 

Chemistry For Engineering Students
ISBN: 9780357026991
4th Edition
Authors: Lawrence S. Brown, Tom Holme
Question Posted:
Students also viewed these Sciences questions
-
Determine A in the indicated figures. Fig. 2.40 (c) B 4 66 A (c) 4 C
-
Using values from the table of standard reduction potentials, calculate the cell potentials of the following cells. (a) (b) (c) Ga(s) | Ga+ (aq) || Ag+ (aq) | Ag(s)
-
Use the table of standard reduction potentials (Appendix M) to calculate r G for the following reactions at 298 K. Data given in Appendix M (a) 3 Cu(s) + 2NO3(aq) + 8 H+ (aq) 3 Cu+ (aq) + 2 NO(g) +...
-
Data for Sabanci Holding can be found in the table below. Theincome statement items correspond to revenues or costs during theyear ending in either 2018 or 2019. The balance sheet itemscorrespond t 2...
-
List six general techniques that are effective in persuasion.
-
Ann obtained this list of apartments. a. Use linear regression analysis to determine if there is a correlation between the square footage and the monthly rent. b. Determine the regression equation....
-
Im like the quarterback of the team. I manage 250 accounts, and anything from billing issues, to service issues, to selling the products. Im really the face to the customer, says Alison Capossela, a...
-
Refer to Data Set 6 in Appendix B and use the paired data consisting of brain volume (cm3) and IQ score. Use the data from Appendix B to testfor rank correlation with a 0.05 significance level.
-
Required information Use the following information for the Quick Study below. (Algo) [The following information applies to the questions displayed below.] Trey Monson starts a merchandising business...
-
Use the Nernst equation to calculate the cell potentials of the following cells at 298 K. (a) 2 Ag+ (aq)(0.50 M) + Ni(s)2 Ag(s) + Ni+ (aq)(0.20 M) (b) Cu(s) + PtCl (aq) (0.10 M) Cu+ (aq) (0.20 M)...
-
Bank 1 and Bank 2 are considering entering a compatibility agreement that would permit the users of each banks ATMs access to the other banks ATMs. Bank 1 has a network of branches and automated...
-
Dene Company uses a periodic inventory system and its accounting records include the following inventory information for the month of July: A physical inventory count determined that 620 units were...
-
Begin by locating a scholarly article regarding the effects of increases on minimum wages and discuss its findings. Does a minimum wage increase lead to reductions in employment, or is the overall...
-
Pick an organization (or you can use an example that has professional relevance), and use the DAC framework to accomplish the following objectives. Provide background and a problem statement of the...
-
Woodruff Company is currently producing a snowmobile that uses five specialized parts. Engineering has proposed replacing these specialized parts with commodity parts, which will cost less and can be...
-
What was Newell's organizational culture like before acquiring Rubbermaid? What was the quadrant? Is the perspective short or long term, inside or outside focused, interested in people, process, or...
-
(7.47 of Koretsky 2nd edition) A binary mixture of species 1 and 2 can be described by the following equation of state: P = a 0 VoT The pure species coefficients are given by: amix y1a1a2Y2 with the...
-
What are the prerequisites for such a scheme to work successfully?
-
Transform the while loop from the previous exercise into an equivalent for loop (make sure it produces the same output).
-
Determine the maximum normal stress developed in the bar when it is subjected to a tension of P = 2 kip. 0.125 in. 1.875 in. 1.25 in. r = 0.25 in. 0.75 in.
-
Determine the maximum axial force P that can be applied to the bar. The bar is made from steel and has an allowable stress of Ï allow = 21 ksi. 0.125 in. 1.875 in. 1.25 in. r = 0.25 in. 0.75 in.
-
The A-36 steel plate has a thickness of 12 mm. If Ï allow = 150 MPa, determine the maximum axial load P that it can support. Calculate its elongation, neglecting the effect of the fillets. r= 30...
-
You would like to have a balance of $600,000 at the end of 15 years from monthly savings of $900. If your returns are compounded monthly, what is the APR you need to meet your goal?
-
Explain the importance of covariance and correlation between assets and understanding the expected value, variance, and standard deviation of a random variable and of returns on a portfolio.
-
On August 1 , 2 0 2 3 , Mark Diamond began a tour company in the Northwest Territories called Millennium Arctic Tours. The following occurred during the first month of operations: Aug. 1 Purchased...

Study smarter with the SolutionInn App


