Calculate the value of the solubility product constant for PbSO 4 from the half-cell potentials.
Question:
Calculate the value of the solubility product constant for PbSO4 from the half-cell potentials.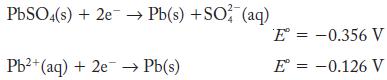
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
E 0073 V ...View the full answer

Answered By

Bree Normandin
Success in writing necessitates a commitment to grammatical excellence, a profound knack to pursue information, and a staunch adherence to deadlines, and the requirements of the individual publication. My background comprises writing research projects, research meta-analyses, literature reviews, white paper reports, multimedia projects, reports for peer-reviewed journals, among others. I work efficiently, with ease and deliver high-quality outputs within the stipulated deadline. I am proficient in APA, MLA, and Harvard referencing styles. I have good taste in writing and reading. I understand that this is a long standing and coupled with excellent research skills, analysis, well-articulated expressions, teamwork, availability all summed up by patience and passion. I put primacy on client satisfaction to gain loyalty, and trust for future projects. As a detail-oriented researcher with extensive experience surpassing eight years crafting high-quality custom written essays and numerous academic publications, I am confident that I could considerably exceed your expectations for the role of a freelance academic writer.
5.00+
7+ Reviews
21+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
Calculate the solubility product constant for copper(II) iodate, Cu(IO3)2. The solubility of copper(II) iodate in water is 0.13 g/100 mL.
-
Use the data in Appendix 2B and the fact that, for the half-reaction F 2 (g) + 2 H + (aq) + 2 e 2 HF(aq), E = 13.03 V, to calculate the value of K a for HF. 2B STANDARD POTENTIALS AT 25 C...
-
(a) Use data from Appendix 2B to calculate the solubility product of Hg 2 Cl 2 . (b) Compare this number with the value listed in Table 6I.1 and comment on any difference. TABLE 61.1 Solubility...
-
Read the following facts and then choose the correct option below: X acts as an interpreter in a transaction where Y wants to buy stolen goods from Z. X's conduct is considered to be that of a/an: a....
-
Consider the following grooves, each of width W, that have been machined from a solid block of material. (a) For each case obtain an expression for the view factor of the groove with respect to the...
-
Bronfenbrenner Co. uses a standard cost system for its single product in which variable overhead is applied on the basis of direct labor hours. The following information is given: Required: Compute...
-
The balance sheets and additional information relating to Pennylane Ltd are given below. Prepare a cash flow statement for Pennylane Ltd for the year ended 31 December 2003 as required under FRS 1...
-
Lynn Rogers (who just turned 30) currently earns $60,000 per year. At the end of each calendar year, she plans to invest 10% of her annual income in a tax-deferred retirement account. Lynn expects...
-
Please solve. Problem 3 (8 points): Assume the term structure of interest rates is flat and consider a 1-factor model with a factor equal to that interest rate. Assume also the current interest rate...
-
What is the voltage of a concentration cell of Fe 2+ ions where the concentrations are 0.0025 and 0.750 M ? What is the spontaneous reaction?
-
Calculate the value of the solubility product constant for Cd(OH) 2 from the half-cell potentials.
-
Refer to the information in E9-3, except assume that Mustafa hopes to make a gain on the bonds as interest rates are expected to fall. Mustafa accounts for the bonds at fair value with changes in...
-
A cube of side L = 2.5 m lies in a region where the electric field is given by = [2(+22 + 5.2) E 3. X L (a) What is the flux through the left face of the cube? N.m/c (b) What is the flux through the...
-
Example. A fair die is rolled until 6 comes up for the 3rd time. What is the probability of obtaining 6 for the 3rd time at 10th trial? Solution.
-
10 Global Financial currently has 8 million common shares outstanding. Over the last three years, the company experienced a substantial sell-off of shares as the earnings per common share fell and...
-
Discuss income replacement versus needs analysis approaches to estimating family insurance needs. 2. Discuss the issues surrounding key employee insurance. 3. Discuss the adverse tax consequences of...
-
Consider an existing capacity resource such as a machine whose capacity is measured in hours it can be realistically operated in a year. In some months, the machine is idle for a few hours because of...
-
Multiple Choice Questions 1. A parent company buys bonds on the open market that had been previously issued by its subsidiary. The price paid by the parent is less than the book value of the bonds on...
-
A container holds 2.0 mol of gas. The total average kinetic energy of the gas molecules in the container is equal to the kinetic energy of an 8.0 10-3-kg bullet with a speed of 770 m/s. What is the...
-
A proposed design for a part of a seawall consists of a rectangular solid weighing 3840 lb with dimensions of 8.00 ft by 4.00 ft by 2.00 ft. The 8.00-ft side is to be vertical. Will this object float...
-
A platform is being designed to support some water pollution testing equipment. As shown in Fig. 5.31, its base is 36.00 in wide, 48.00 in long, and 12.00 in high. The entire system weighs 130 lb,...
-
A block of wood with a specific weight of 32 lb/ft 3 is 6 by 6 by 12 in. If it is placed in oil (sg = 0.90) with the 6 by 12-in surface parallel to the surface of the oil, would it be stable?
-
A built - in refrigerator is considered:real property that won't transferpersonal property that won't transferpersonal property that will transferreal property that will transfer
-
You may find it helpful to review Chapter 7 , Section 7 . 1 "Pressure in fluids" and Study session 1 1 . 1 "Fluid properties and hydrostatics" before attempting this question. A hydraulic accumulator...
-
A firm with flexible policy of short-term financing will have Option A - lower carrying costs. Option B - All of the listed choices Option C - some investments in marketable equity securities. Option...

Study smarter with the SolutionInn App


