Write the expected rate law and molecularity for each of the following elementary reactions in the gas
Question:
Write the expected rate law and molecularity for each of the following elementary reactions in the gas phase.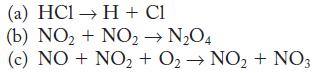
Strategy
The rate law for an elementary reaction is derived directly from its stoichiometry. The molecularity is the sum of the exponents of the concentrations in the elementary step.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:





