From the relative atomic orbital and molecular orbital energies depicted in Fig. 3.32, describe the character as
Question:
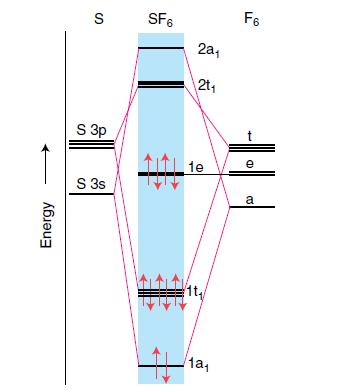
From the relative atomic orbital and molecular orbital energies depicted in Fig. 3.32, describe the character as mainly F or mainly S for the frontier orbitals e (the HOMO) and 2t (the LUMO) in SF6. Explain your reasoning.
Figure 3.32.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Inorganic Chemistry
ISBN: 9780198768128
7th Edition
Authors: Mark Weller, Tina Overton, Jonathan Rourke
Question Posted:





