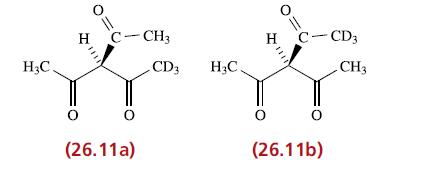
The rate of racemization of [CoL 3 ] where HL = 26.11a is approximately the same as
Question:
The rate of racemization of [CoL3] where HL = 26.11a is approximately the same as its rate of isomerization into [CoL'3] where HL' =26.11b. What can you deduce about the mechanisms of these reactions?
Transcribed Image Text:
H₂C. H O C-CH3 O (26.11a) CD3 H₂C H O C-CD3 (26.11b) CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 85% (7 reviews)
Based on the given information we can deduce that the mechanisms of racemization and isomerization of CoL3 are likely to be similar or closely related ...View the full answer

Answered By

Deepak Sharma
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The chemical shifts of the 1H nuclei in 2,2-dimethylpropane and TMS are 0.95 and 0.0, respectively. From these data, what can you deduce about the relative electronegativities of carbon and silicon?
-
The rate constants for racemization (kr) and dissociation (k d ) of [FeL 3 ] 4 (H 2 L = 26.12) at several temperatures, T, are given in the table. (a) Determine for each reaction. (b) What can you...
-
Kinesin-1 contains two identical heavy chains and therefore has two identical motor domains. In contrast, kinesin-5 contains four identical heavy chains. Electron microscopic analysis of...
-
Calculate the density of N2 at STP, (a) using the ideal-gas law and (b) using the molar volume and molar mass of N2. How do the densities compare?
-
The balance sheet for NuBone Corporations New Products Division showed invested assets of $200,000 at the beginning of the year and $300,000 at the end of the year. During the year, the divisions...
-
For the rotational system shown in Figure P2.25, write the equations of motion from which the transfer function, G(s) = 1 (s)/T(s), can be found. Figure P2.25 T(t) 0,(1) N J. K D N2 N3 J2 N4 JL. J4...
-
Describe the benefits that intangible assets provide to a company.(Appendix)
-
Methanol is synthesized from carbon monoxide and hydrogen in a catalytic reactor. The fresh feed to the process contains 32.0 mole% CO. 64.0% H2 and 4.0% N2. This stream is mixed with a recycle...
-
I need help with letter E&F please
-
(a) The reaction: occurs by a dissociative mechanism and the first order rate constants, k 1 , vary with the nature of substituent X as follows: CO 3 P(OPh) 3 nBu 3 : Comment on these data. (b) The...
-
Rationalize the observation that when the reaction: is carried out in H 2 ( 18 O), the water in the complex contains equal proportions of H 2 ( 18 O) and H 2 ( 16 O). [Co(NH3)4 (CO3)]* [HO]*, HO 13+...
-
Using the Shewhart charting methods described in Figure 2.4 in Chapter 2, analyze the performance of a check-clearing process. The standard time to clear a check is six seconds. The allowed variation...
-
An apartment building owner is interested in a mortgage from a Canadian bank. What is the largest monthly mortgage payment that would be allowed given the following facts: square footage of...
-
Use NPV to analyze the decision to purchase the new machine. A) Identify and label each inflows/outflow, don't just present a single inflow/outflow number for each year. B) Assuming a discount rate...
-
What is the breakeven sales in peso? How much is the margin of safety The number of units that must be sold if the company wants to earn an income after tax of 150,000. Assuming an income tax rate is...
-
A local TV repairs shop uses 36,000 units of a part each year (A maximum consumption of 100 units per working day). It costs P20 to place and receive an order. The shop orders in lots of 400 units....
-
ONYX Corporation, began its operation on January 1, 2021. It produces Product A that sell for P13.50 per unit. During 2021 150,000 units were produced and 15,000 units were left unsold in 2021....
-
Let g and h be real-valued functions defined on R for which «R g(x)dμ and «R h(x)dμ are finite, where μ is a (Ï-finite) measure in R. Then...
-
The Dow Jones Industrial Average reached a high of $ 7801.63 on December 29, 1997. Recall from Example 18.4 that it reached a high of $ 1003 on November 14, 1972. The Consumer Price Index for...
-
Draw a plausible mechanism for each of the following reactions: (a) (b) [H,SO4] -H20 [H,SO4] -H20
-
Draw a plausible mechanism for each of the following reactions: (a) (b) HO HO- [H2SO4] -H20
-
Predict the product of each of the following reactions: (a) (b) [H2SO4] excess MeOH -? -H20 [H,SO4] -H20
-
On January 1 , Boston Enterprises issues bonds that have a $ 1 , 2 0 0 , 0 0 0 par value, mature in 2 0 years, and pay 9 % interest semiannually on June 3 0 and December 3 1 . The bonds are sold at...
-
Analyze the process costing system used by General Motors (GM) in its automobile manufacturing plants. Last year, one of GM's plants incurred total costs of $8,000,000. During the same period, the...
-
Kevin was hired as the Chief Executive Officer (CEO) of Iota Corporation, a privately-owned company in the energy sector, effective February 1, 2022. Throughout the fiscal year 2023, Iota Corporation...

Study smarter with the SolutionInn App


