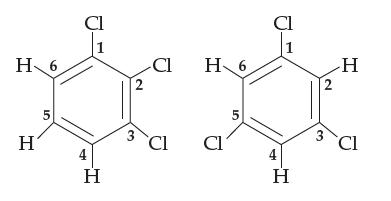
Consider 1,2,3-trichlorobenzene and 1,3,5-trichlorobenzene, both shown below (the carbon atoms are numbered as shown). Both are flat
Question:
Consider 1,2,3-trichlorobenzene and 1,3,5-trichlorobenzene, both shown below (the carbon atoms are numbered as shown). Both are flat molecules with 120° bond angles, yet one of them is polar and the other nonpolar. Which is which, and why? For the polar one, indicate which part of the molecule is δ+ and which is δ–.
Transcribed Image Text:
H 6 H 5 Cl 1 H 2 3 -Cl Cl H 6 Cl 5 Cl H 2 3 H Cl
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (6 reviews)
123trichlorobenzene is polar while 135trichlorobenzene is nonpolar To determine the polarity of a mo...View the full answer

Answered By

Mustafa olang
Please accept my enthusiastic application to solutionInn. I would love the opportunity to be a hardworking, passionate member of your tutoring program. As soon as I read the description of the program, I knew I was a well-qualified candidate for the position.
I have extensive tutoring experience in a variety of fields. I have tutored in English as well as Calculus. I have helped students learn to analyze literature, write essays, understand historical events, and graph parabolas. Your program requires that tutors be able to assist students in multiple subjects, and my experience would allow me to do just that.
You also state in your job posting that you require tutors that can work with students of all ages. As a summer camp counselor, I have experience working with preschool and kindergarten-age students. I have also tutored middle school students in reading, as well as college and high school students. Through these tutoring and counseling positions, I have learned how to best teach each age group. For example, I created songs to teach my three-year-old campers the camp rules, but I gave my college student daily quizzes to help her prepare for exams.
I am passionate about helping students improve in all academic subjects. I still remember my excitement when my calculus student received her first “A” on a quiz! I am confident that my passion and experience are the qualities you are looking for at solutionInn. Thank you so much for your time and consideration.
4.80+
2+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
A local manufacturing company produces heating and colling appliances for home and industrial applications. These include refrigerators, water coolers, water heaters, air conditioners, sandwich...
-
Consider the tensile stress-strain diagrams in Figure 6-28 labeled 1 and 2 and answer the following questions. These diagrams are typical of metals. Consider each part as a separate question that has...
-
For each of the following organic molecules draw a Lewis structure in which the carbon atoms are bonded to each other by single bonds: (a) C2H6, (b) C4H10, (c) C5H12. For (b) and (c), show only...
-
The OrbitTrack Company specializes in developing and selling a wide range of high-quality scooters. Sales representatives report that there is a growing demand for racing scooters. OrbitTrack's...
-
Spaulding Manufacturing Company has determined the cost of manufacturing a unit of product as follows, based on normal production of 100,000 units per year: Direct materials . . . . . . . . . . . . ....
-
Russell Inc. had sales of $2,200,000 for the first quarter of 2017. In making the sales, the company incurred the following costs and expenses. Prepare a CVP income statement for the quarter ended...
-
Jamie Lee is considering a used vehicle but cannot decide where to begin her search. Using Personal Financial Planner Sheet 34, Transportation Needs, and the internet for your research, evaluate two...
-
Suppose the federal government requires beer drinkers to pay a $2 tax on each case of beer purchased. (In fact, both the federal and state governments impose beer taxes of some sort.) a. Draw a...
-
Assessing the existence of a contract: No written sales agreement Shoe Manufacturer S holds products available to ship to customers before the end of its current fiscal year. Shoe Shop T places an...
-
Cyclohexane, formula C 6 H 12 , is often drawn as a puckered (not flat) ring as shown below (the purple and blue atoms are the hydrogens). Explain how drawing it as a puckered ring makes it...
-
There are exceptions to the predictions of VSEPR. Consider CH 3 , known as a methyl radical. (a) Create a dot diagram for the methyl radical. How is it fundamentally different from other dot diagrams...
-
Why all the flap about atomic bombs? The potential for death is always with us. Of course, if you just want something to worry about, go ahead. Franklin D. Roosevelt said it: The only thing we have...
-
(ii) State Wilkie's updating equation in respect of the force of inflation and explain carefully what each of the components of the equation represents. State also which type of time series process...
-
Compute the double integral D x y dA over the domain D indicated as 0 x 5, x y 2x + 3. (Use symbolic notation and fractions where needed.) f(x, y) A = D
-
4. (10 points) A researcher believes that length of time spent listening to classical music increases memory for previously learned material. She has 4 groups of 5 subjects listen to either 10 min.,...
-
We find a binary system consisting of a 1 solar mass star, still in its main sequence phase, and a white dwarf. Assume both stars formed at the same time and that they did not significantly influence...
-
The equity sections from Atticus Group's 2015 and 2016 year-end balance sheets follow. Stockholders Equity (December 31, 2015) Common stock $6 par value, 50,000 shares authorized, 35,000 shares...
-
Find the slope of the tangent line to the curve of intersection of the vertical plane x - (3y + 2(3 - 1 = 0 and the surface z - x2 + y2 at the point (1, 2, 5).
-
Planning: Creating an Audience Profile; Collaboration: Team Projects. Compare the Facebook pages of three companies in the same industry. Analyze the content on all available tabs. What can you...
-
Find the result of operating with (1/r 2 ) (d /dr) (r 2 d /dr) + 2/r on the function Ae br . What must the values of A and b be to make this function an eigenfunction of the operator?
-
Normalize the set of functions n (θ) = e inθ , 0 ¤ θ ¤ 2Ï. To do so, you need to multiply the functions by a so-called normalization constant...
-
Show that the set of functions n () = e in , 0 2is orthogonal if n is an integer. To do so, you need to 2 0 * m () n () = d = 0 for m n if n and m are intergers.
-
ABC company makes turbo-encabulators, customized to satisfy each customers order. They split overhead into five pools, each with its own activity driver (direct labor for manufacturing, direct labor...
-
Variable manufacturing overhead becomes part of a unit's cost when variable costing is used.Group of answer choicesTrueFalse
-
Santa Fe Corporation has computed the following unit costs for the year just ended:Direct Material used $23Direct Labor $18Fixed selling and administrative cost $18Variable manufacturing overhead...

Study smarter with the SolutionInn App


