In the course of the synthesis of the sex attractant of the grape berry moth, both the
Question:
In the course of the synthesis of the sex attractant of the grape berry moth, both the cis and trans isomers of the following alkene were needed.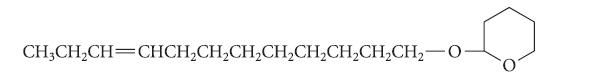
(a) Outline a synthesis of the cis isomer of this alkene from the following alkyl halide and any other organic compounds.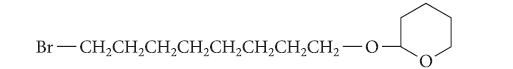
(b) Outline a synthesis of the trans isomer of the same alkene from the same alkyl halide and any other organic compounds.
Transcribed Image Text:
CH,CH,CH=CHCH,CH,CH,CH,CH,CH,CH,CH,−0
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
a When a cisalkene is called for catalytic hydrogenation of an alkyne is the preferred rou...View the full answer

Answered By

Lamya S
Highly creative, resourceful and dedicated High School Teacher with a good fluency in English (IELTS- 7.5 band scorer) and an excellent record of successful classroom presentations.
I have more than 2 years experience in tutoring students especially by using my note making strategies.
Especially adept at teaching methods of business functions and management through a positive, and flexible teaching style with the willingness to work beyond the call of duty.
Committed to ongoing professional development and spreading the knowledge within myself to the blooming ones to make them fly with a colorful wing of future.
I do always believe that more than being a teacher who teaches students subjects,...i rather want to be a teacher who wants to teach students how to love learning..
Subjects i handle :
Business studies
Management studies
Operations Management
Organisational Behaviour
Change Management
Research Methodology
Strategy Management
Economics
Human Resource Management
Performance Management
Training
International Business
Business Ethics
Business Communication
Things you can expect from me :
- A clear cut answer
- A detailed conceptual way of explanation
- Simplified answer form of complex topics
- Diagrams and examples filled answers
4.90+
46+ Reviews
54+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The P/E ratio on the S&P 500 Index for 1998and 1999 was 30 or higher. Other things equal,would this indicate a good time to buy stocks fora multi-year holding period, or not?
-
Your firm has selected you to develop and assess the control risk over shipping and billing functions of ABC Company. The audit manager wishes to rely on control risk assessment at a low level. In...
-
Dickens, Kristen, is enrolled as a doctoral student in the Counselor Education at the University of New Orleans. She is a registered counselor intern in the state of Louisiana and works at a...
-
Why is an investment portfolio containing a mix of stocks and bonds less risky than one containing a single asset class? Because the markets for stocks and bonds tend to move in the same direction at...
-
Following is a bank reconciliation for Fez's Sandwich Shop for May 31, 2010. Because of limited funds, Fez's employed only one accountant who was responsible for receiving cash, recording receipts...
-
The article "A Thin-Film Oxygen Uptake Test for the Evaluation of Automotive Crankcase Lubricants" (Lubric. Engr., 1984: 75-83) reported the following data on oxidation-induction time (min) for...
-
Suppose W > 0, C, and satisfy the intertemporal budget constraint (13.38). Define the consumption-reinvested wealth process W by (13.43). (a) Show that W satisfies the intertemporal budget...
-
An inexperienced accountant prepared this condensed income statement for Hight Company, a retail firm that has been in business for a number of years. HIGHT COMPANY Income Statement For the Year...
-
The following 2 mutually exclusive projects ( Project A and Project B ) are available : Year / s Cash Flows ( A ) Cash Flows ( B ) 0 - 2 0 0 0 0 0 - 2 0 0 0 0 1 1 8 0 0 0 1 0 0 0 0 2 2 8 0 0 0 9 0 0...
-
Propose a synthesis of 4,4-dimethyl-2-pentyne from an alkyl halide and an alkyne.
-
Explain why graduate student Choke Fumely, in attempting to synthesize 4,4-dimethyl-2-pentyne using the reaction of with tert-butyl bromide, obtained none of the desired product. What product did he...
-
You are an adviser to the Indian government. Until now, government policy in India has been to severely limit imports into India, resulting also in a low level of Indian exports. The government is...
-
Q10: Region ( experienced compressive stresses and has a than the rest of the bracket. Region ( ) experienced tension stresses and has a of the bracket. Deep Drawing and Stretch Forming width (into...
-
A sample of 1500 computer chips revealed that 32% of the chips do not fail in the first 1000 hours of their use. The company\'s promotional literature claimed that above 29% do not fail in the first...
-
The 75 lb block is released from rest 5 ft above the plate. Determine the compression of each spring when the block momentarily comes to rest after striking the plate. Neglect the mass of the plate....
-
Indiana Soy Products (OSP) buys soybeans and processes them into other soy products. Each ton of soybeans that OSP purchases for $250 can be converted for an additional $180 into 675 lbs of soy meal...
-
The 2025 Annual Report of Splish International contains the following informatio (in millions) June 29, 2025 June 27, 2024 Total assets $1,545 $1,502 Total liabilities 989 1,060 Net sales 2,800 2.971...
-
Let f(x + y) = f(x) + f(y) for all x and y and suppose that f is continuous at x = 0. (a) Prove that f is continuous everywhere. (b) Prove that there is a constant in such that f (t) = mt for all t...
-
Danielle has an insurance policy with a premium of $75 per month. In September she is in an accident and receives a bill worth $2990 for the repair of her own property. Her deductible is $250 and her...
-
Addition of HC1 to 1-methoxycyclohexene yields 1-chloro-1-methoxycyclo- hexane as the sole product. Use resonance structures to explain why none of the other regioisomer isformed. HCI CI CI
-
Hydrocarbon A, C 10 H 14 , has a UV absorption at ? max = 236 nm and gives hydrocarbon B, C 10 H 18 , on catalytic hydrogenation. Ozonolysis of A followed by zinc/acetic acid treatment yields the...
-
Adiponitrile, a starting material used in the manufacture of nylon, can be prepared in three steps from 1, 3-butadiene. How would you carry out thissynthesis? 3 steps H2C=CHCH=CH2 N=CCH2CH2CH2CH2C=N...
-
Chapter o Homew ebook 50,000-unit production quantity: $ 227,049 7 70,000-unit production quantity: $ 66,751 d. In addition to mean profit, what other factors should FTC consider in determining a...
-
Diamond makes downhill ski equipment. Assume that comic has offered to produce ski poles for Diamond for $20 per pair Diamond needs 200,000 pairs of poles per period Diamond can only avoid 5150,000...
-
17? Which of the following statement is true Select one: a. All evidence must have the same level of reliability b. All evidence must have the same level of persuasiveness C. All are false d....

Study smarter with the SolutionInn App


