Question: In each case below, identify the acid and the base. Then draw the curved arrows showing a proton transfer reaction. Draw the products of that
In each case below, identify the acid and the base. Then draw the curved arrows showing a proton transfer reaction. Draw the products of that proton transfer, and then predict the position of equilibrium.
(a)
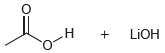
(b)
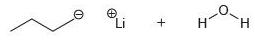
(c)
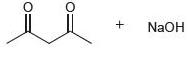
LIOH
Step by Step Solution
3.34 Rating (184 Votes )
There are 3 Steps involved in it
a b c ... View full answer

Get step-by-step solutions from verified subject matter experts


