Question: Indicate whether you would use sodium ethoxide or potassium tert-butoxide to achieve each of the following transformations: a. b. c. d. Br Br
Indicate whether you would use sodium ethoxide or potassium tert-butoxide to achieve each of the following transformations:
a.

b.
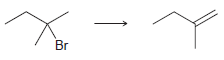
c.
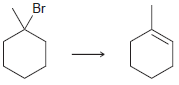
d.
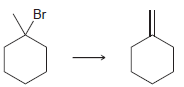
Br Br
Step by Step Solution
★★★★★
3.40 Rating (156 Votes )
There are 3 Steps involved in it
1 Expert Approved Answer
Step: 1 Unlock

a The Zaitsev product is desired so sodium hydroxide shoul... View full answer

Question Has Been Solved by an Expert!
Get step-by-step solutions from verified subject matter experts
Step: 2 Unlock
Step: 3 Unlock


