Question: Using bromobenzene and ethylene oxide as your only sources of carbon, show how you could prepare trans- 1, 2-diphenyloxirane (a racemic mixture of enantiomers). +
Using bromobenzene and ethylene oxide as your only sources of carbon, show how you could prepare trans- 1, 2-diphenyloxirane (a racemic mixture of enantiomers).
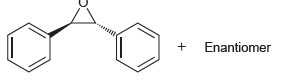
+ Enantiomer
Step by Step Solution
★★★★★
3.42 Rating (161 Votes )
There are 3 Steps involved in it
1 Expert Approved Answer
Step: 1 Unlock

Br 1 Mg 2 3 HO MCP... View full answer

Question Has Been Solved by an Expert!
Get step-by-step solutions from verified subject matter experts
Step: 2 Unlock
Step: 3 Unlock


