0.0050 mol of gas undergoes the process 1?? 2 ?? 3 shown in Figure P16.38.? What are...
Question:
0.0050 mol of gas undergoes the process 1?? 2 ?? 3 shown in Figure P16.38.?
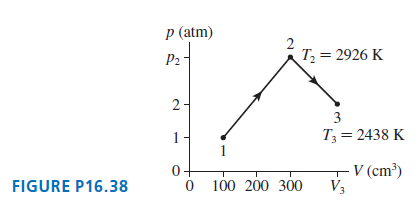
What are
(a) Temperature T1,
(b) Pressure p2,
(c) Volume V3?
Transcribed Image Text:
p (atm) P2 - T = 2926 K 3 T3 = 2438 K 1 0+ V (cm³) V3 FIGURE P16.38 100 200 300
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 77% (18 reviews)
Model Assume the gas is an ideal gas Solve a Using the i...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Physics for Scientists and Engineers A Strategic Approach with Modern Physics
ISBN: 978-0133942651
4th edition
Authors: Randall D. Knight
Question Posted:
Students also viewed these Physics questions
-
0.0040 mol of gas undergoes the process shown in FIGURE EX18.35. a. What type of process is this? b. What are the initial and final temperatures in °C? p (atm) 3- 2- V (cm) 300 0+ 100 200 FIGURE...
-
0.020 mol of gas undergoes the process shown in FIGURE EX18.37. a. What type of process is this? b. What is the final temperature in °C? c. What is the final volume V 2 ? p (atm) 3- 2- 1- V (cm)...
-
8.0 g of helium gas follows the process 1 ?? 2 ?? 3 shown in FIGURE P18.60. Find the values of V 1 , V 3 , p 2 , and T 3 . P2- 657C - Isothermal 2 atm - 37C- 3 0+ -v V1 V3 FIGURE P18.60
-
e) Based on your investigation of the data set create two well-formatted tables that illustrate some important features of the data. Your tables should have informative labels and a readable number...
-
John Curran and his wife own a construction company, and they want to set up two new corporations that will be owned by their Roth IRAs. The two Roth-owned corporations will then enter into contracts...
-
As a long-term investment, Painters' Equipment Company purchased 20% of AMC Supplies Inc.'s 540,000 shares for $620,000 at the beginning of the fiscal year of both companies. On the purchase date,...
-
(Earnings per Share) At December 31, 2003, Shiga Naoya Corporation had the following stock outstanding. 10% cumulative preferred stock, $100 par, 107,500 shares $10,750,000 Common stock, $5 par,...
-
Sallys Software Inc. is a rapidly growing supplier of computer software to the Sarasota area. Sales for the last 5 years, 2009 to 2013, are given below. Sales Year ($ millions) 2009 .........1.1 2010...
-
During the year, a firm purchased $ 1 4 4 , 0 0 0 of merchandise and paid freight charges of $ 1 2 , 6 0 0 . If the total purchases returns and allowances were $ 9 , 2 0 0 and purchases discounts...
-
Use the accompanying data of the retail survey to state some examples of interesting hypothesis tests by proposing null and alternative hypotheses. Click the icon to view the retail survey data. If...
-
iRobot designs and manufactures robots for consumer, commercial, and military use. For the fiscal year ended January 2, 2016, the company reported the following on its balance sheet and income...
-
An ideal gas starts with pressure p 1 and volume V 1 . Draw a pV diagram showing the process in which the gas undergoes an isochoric process that doubles the pressure, then an isobaric process that...
-
(a) How does a system of standard costing enable a business to operate on the principle of management by exception? (b) Some of the following materials and labour variances have been wrongly...
-
n1 = 15, n2 = 18, S = 280, H1: m1 > m2. Exercises 57 present sample sizes and the sum of ranks for the rank-sum test. Compute S, S, and the value of the test statistic z. Then find the P-value.
-
n1 = 25, n2 = 32, S = 850, H1: m1 m2. Exercises 57 present sample sizes and the sum of ranks for the rank-sum test. Compute S, S, and the value of the test statistic z. Then find the P-value.
-
Evaluate the matrix element $\left\langle j_{1} j_{2} J\left|T_{k q}(1) ight| j_{1}^{\prime} j_{2}^{\prime} J^{\prime} ightangle$, where the tensor operator $T_{k q}(1)$ operates only on the part of...
-
Mark Gold opened Gold Roofing Service on April 1. Transactions for April are as follows: 1 Gold contributed \(\$ 15,000\) of his personal funds in exchange for common stock to begin the business. 2...
-
n1 = 20, n2 = 30, S = 400, H1: m1 < m2. Exercises 57 present sample sizes and the sum of ranks for the rank-sum test. Compute S, S, and the value of the test statistic z. Then find the P-value.
-
Use Fig. 2.134. Given that AB = 4, BC = 4, CD = 6, and ADC = 53, find the indicated angle and lengths. BE Fig. 2.134 A B E C D
-
You are interested in investing and are considering a portfolio comprised of the following two stocks. Their estimated returns under varying market conditions are provided: (note: it is difficult to...
-
Consider a house in a northern location that needs a total of 100 GJ of thermal energy over a winter season from November through March. Assume that the house has a ground source heat pump that...
-
Compute the draw down coefficient (32.11) in a simple model where the geothermal reservoir is a large cylindrical volume with height h much smaller than the radius r (see Figure 32.17) Assume that...
-
Consider a geothermal reservoir (see Figure 32.17) At a depth of 1.5 km, modeled as a cylindrical volume of radius r = 5 km and height h = 500m, with pressure at the periphery of p r 15 MPa,...
-
Difference between Operating Leverage and Financial Leverage
-
bpmn diagram for misc purchases
-
You have $55,000. You put 15% of your money in a stock with an expected return of 10%, $38,000 in a stock with an expected return of 18%, and the rest in a stock with an expected return of 22%. What...

Study smarter with the SolutionInn App


