Answered step by step
Verified Expert Solution
Question
1 Approved Answer
A system consists of 1 mole of perfect gas with an initial temperature of 0 C and an initial pressure of 1 atm. This system
A system consists of 1 mole of perfect gas with an initial temperature of 0 C and an initial pressure of 1 atm. This system was subjected to the following processes, each performed under reversible conditions:
(i) A 5-fold increase in volume at constant temperature, (ii) Subsequently, a 30-fold increase in pressure under no-heat conditions, (iii) Subsequent return to the origin along a line on the P-V diagram. (a) Calculate the work done by the system in step (iii) (b) Calculate the total heat flow in the cyclic process.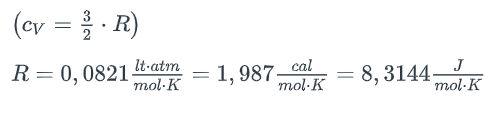
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


