Draw resonance structures for this anion. Remember, sulfur can have 10 or even 12 electrons in its
Question:
Draw resonance structures for this anion. Remember, sulfur can have 10 or even 12 electrons in its valence shell.
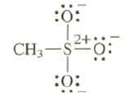
Transcribed Image Text:
:0: 24 CH₂-S2+0 :0:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 81% (11 reviews)
0 21 CH3SO 0 ...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Draw resonance structures for each of the following compounds: a. b. c. d. e. f. g. h. i. j. N.
-
Draw resonance structures for each of the following radicals: (a) (b) (c) (d)
-
Draw resonance structures for each of the following anions. a. b. c.
-
CPA firm brings in a yoga instructor during the tax busy season to help relieve stress of the employees. Which is true about the CPA firm's ability to take a deduction for the yoga instructor's...
-
Frosty Menu Company produces cases of frozen food. During April, the company produced 1450 cases of food and incurred the following actual costs: Variable overhead...
-
What is monetary policy?
-
The following quotations are available to you. (You may either buy or sell at the stated rates.) Won714.00/S$ HK$4.70/S$ Singapore bank: Singapore dollar quote for Korean won Hong Kong bank: HKS...
-
The following information was taken from the financial statements of Tolbert Inc. for December 31 of the current fiscal year: Common stock , $20 par (no change during the year).... $10,000,000...
-
In 2009, the life expectancy of males in a certain country was 73 years and increasing at a rate of 0.4 year per year. Model this situation by a linear equation; use this equation to estimate the...
-
Based on your answer to question 9, construct a first-quarter 2015cash flow forecast for Westmark Industrial. Westmark Industrials annual income statement and balance sheet for December 31, 2014...
-
Consider H2C = C = CH2. What is the hybridization and geometry at each C? Indicate the bond types for each of the carbon-carbon bonds. Draw the molecule, showing the overall geometry and the p...
-
Phenanthrene has five total resonance structures. One is shown here. Draw the other four. Which carbon-carbon bond of Phenanthrene would your predict to be the shortest? Phenanthrene
-
What is meant by a use of cash?
-
Roask Conference Centre is considering investing in some new audio visual equipment. The required equipment has a 7-year life. Also, some new working capital would be required and would be recovered...
-
Give alot of information about Light OS and Tiny OS. Explain what they are used for and how they will function differently comparing Light OS and Tiny OS. What differences could they make and how...
-
Questions based on southwest data: Make a final recommendation - would you buy or sell? Why? (Hint: With the current volatile market condition, make decisions based on your investment holding period....
-
How do I step by step and where do I Construct a portfolio consisting of the following assets: My chosen cryptocurrency An equity index (e.g., S&P 500, EURO STOXX 50) A fixed-income asset (e.g.,...
-
3. For f(x)= 5x x +4 : find where the function is increasing and where it is decreasing, along any relative extrema.
-
Why is flexible budgeting a more valuable tool for logistics managers than fixed dollar budgeting?
-
Review Exhibit 11.4. Analyze each product on the graph according to the characteristics that influence the rate of adoption. For example, what can you conclude from the data about the relative...
-
Determine whether the series converges or diverges. 00 5 n=1 2n + 4n+ 3
-
Name the following compound, identify each substituent as axial or equatorial, and tell whether the conformation shown is the more stable or less stable chair form (yellow-greenC1):
-
A trisubstituted cyclohexane with three substituents? red, yellow, and blue?undergoes a ring-flip to its alternative chair conformation. Identify each substituent as axial or equatorial, and show the...
-
Glucose exists in two forms having a 36 : 64 ratio at equilibrium. Draw a skeletal structure of each, describe the difference between them, and tell which of the two you think is more stable (red =...
-
Saham dan obligasi sudah begitu mudah diperjualbelikan oleh masyarakat melalui aplikasi handphone, seperti Stockbit, Bibit, Pluang, Mobile banking, dsb. Cukup dengan modal mulai Rp100.000,-...
-
Consider a map marked with the locations of major cities and a table of pairwise distances between those cities. In which cases is each more useful? Choose the best answer. Group of answer choices...
-
Which of the following would NOT generally help increase overall PE's annual returns for a LBO transaction? Exiting the transaction earlier Issuing more debt for a dividend Decreasing capital...

Study smarter with the SolutionInn App


