Question: Reaction of acetone with D 3 O + yields hexadeuterioacetone. That is, all the hydrogen?s in acetone are exchanged for deuterium. Review the mechanism of
Reaction of acetone with D3O+ yields hexadeuterioacetone. That is, all the hydrogen?s in acetone are exchanged for deuterium. Review the mechanism of mercuric ion?catalyzed alkyne hydration, arid then propose a mechanism for this deuterium incorporation.
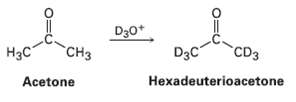
D30* "CH H3C D3C CD3 Acetone Hexadeuterioacetone
Step by Step Solution
★★★★★
3.42 Rating (168 Votes )
There are 3 Steps involved in it
1 Expert Approved Answer
Step: 1 Unlock

24txA4 HC HC HC CH3 enol CH3 CH3 CH3 HC HCC HC ... View full answer

Question Has Been Solved by an Expert!
Get step-by-step solutions from verified subject matter experts
Step: 2 Unlock
Step: 3 Unlock
Document Format (1 attachment)
22-C-O-O-S (47).docx
120 KBs Word File


