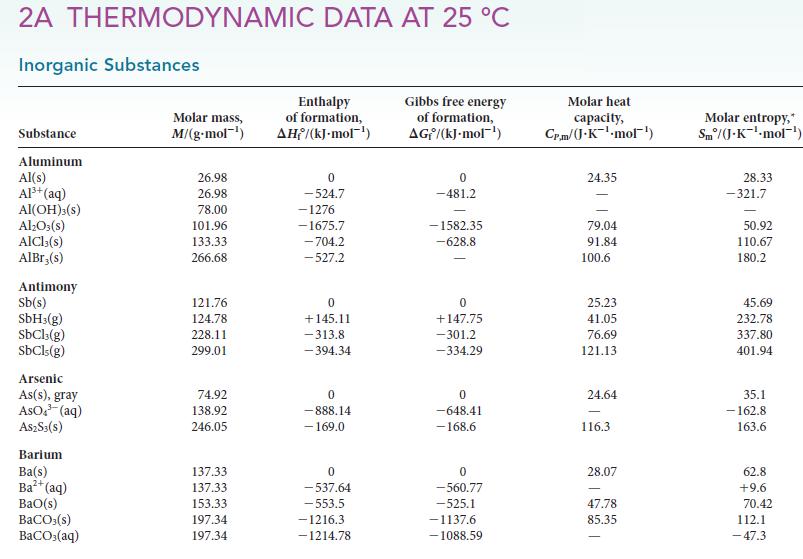
Use standard enthalpies of formation from Appendix 2A to calculate the standard reaction enthalpy for each of
Question:
Use standard enthalpies of formation from Appendix 2A to calculate the standard reaction enthalpy for each of the following reactions:
(a) The final stage in the production of nitric acid:![]()
(b) The industrial synthesis of boron trifluoride:![]()
(c) The formation of a sulfide by the action of hydrogen sulfide on an aqueous solution of a base:![]()
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:





