Ammonia gas can be prepared by the following reaction: If 112 g of CaO and 224 g
Question:
Ammonia gas can be prepared by the following reaction:
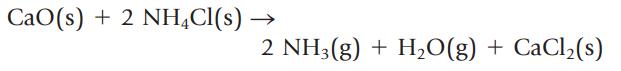
If 112 g of CaO and 224 g of NH4Cl are mixed, the theoretical yield of NH3 is 68.0 g. If only 16.3 g of NH3 is actually obtained, what is its percent yield?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:





