Suppose the exothermic reaction has come to equilibrium. Which of the following are true? (a) Adding O
Question:
Suppose the exothermic reaction
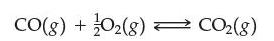
has come to equilibrium. Which of the following are true?
(a) Adding O2(g) will shift the reaction to the left.
(b) Cooling the reaction will shift it to the right.
(c) Cooling the reaction will shift it to the left.
(d) All of the above are true.
Transcribed Image Text:
(8) 10) = (8)' 10 2 OF + CO(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
b Cooling ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
15. Mama Corporation (a U.S. taxpayer) has a wholly-owned sales subsidiary in the Bahamas (Bahamamama Ltd.) that purchases finished goods from its U.S. parent and sells those goods to customers...
-
X Finding Values of an Inverse Function Assume that f is a one-to-one function. -25. (a) If f(2)= 7, find f(7). (b) If f(3) = -1, find f(-1). 26. (a) If f(5) = 18, find f(18). (b) If f(4) = 2, find...
-
In the film, Brain Matters, it showed that researchers, scientists and educators have outlined a set of four brain-boosting processes that will strengthen neural pathways and help children to be...
-
McGuire Industries prepares budgets to help manage the company. McGuire is budgeting for the fiscal year ended January 31, 2021. During the preceding year ended January 31, 2020, sales totaled $9,200...
-
Friar Paint Company makes paint in many different colors; it charges the same price for all of its paint regardless of the color. Recently, Friars chief competitor cut the price of its white paint,...
-
There are 50 people in a class. The teacher wants a random sample of 10 students. She numbers the students consecutively (1, 2, 3, 4, . . . 50) and uses the random number able to sample 10 cases...
-
Suppose MdRd is a martingale and define the risk-neutral probability corresponding to Md. Assume MdXRf is also a martingale. Show that dX X = (r d r f )dt +x dB , where B is a Brownian motion under...
-
Prepare budgetary entries, using general ledger accounts only, for each of the following unrelated situations: a. Anticipated revenues are $10 million; anticipated expenditures and encumbrances are...
-
on june 30, tower company issues 8%, 20-year bonds payable with a face amount of $100,000. The bonds are issues at 95 (this is a great to given the 95% number instead of having to use the tables to...
-
Write full nuclear reactions for Practice Problems 16.10 and 16.11. Data from Practice Problems 16.10 The magnesium isotope 25 12 Mg is the daughter isotope created when a radioactive parent isotope...
-
Suppose the shortcut rules can determine the oxidation state of every atom in a compound except one. How can you find the oxidation state of the remaining atom?
-
A common device used in various applications to clean particle-laden air is the reverse-flow cyclone (Fig. P7120). Dusty air (volume flow rate V and density ) enters tangentially through an opening...
-
Below are incomplete financial statements for Hurricane, Incorporated Required: Calculate the missing amounts. Complete this question by entering your answers in the tabs below. Income Statement Stmt...
-
TBTF Incorporated purchased equipment on May 1, 2021. The company depreciates its equipment using the double-declining balance method. Other information pertaining to the equipment purchased by TBTF...
-
Coco Ltd. manufactures milk and dark chocolate blocks. Below is the information relating to each type of chocolate. Milk Chocolate Selling price per unit $6 Variable cost per unit $3 Sales mix 4 Dark...
-
Data related to 2018 operations for Constaga Products, a manufacturer of sewing machines: Sales volume 5,000 units Sales price $300.00 per unit Variable production costs Direct materials 75.00 per...
-
6. (20 points) Sections 3.1-3.5, 3.7 Differentiate the following functions, state the regions where the functions are analytic. a. cos(e*) b. 1 ez +1 c. Log (z+1) (Hint: To find where it is analytic,...
-
The rate law for the reaction of HO- with tert-butyl bromide to form an elimination product in 75% ethanol 25% water at 30 C is the sum of the rate laws for the E2 and E1 reactions: rate = 7.1 ...
-
The tractor is used to lift the 150-kg load B with the 24-mlong rope, boom, and pulley system. If the tractor travels to the right at a constant speed of 4 m/s, determine the tension in the rope when...
-
Mount McKinley (also called Denali) is the tallest mountain in North America, with a height of 6200 m above sea level. If a person of mass 120 kg walks from sea level to the top of Mount McKinley,...
-
Consider again the roller coaster in Figure P6.32, but now assume the roller coaster starts with a speed of 12 m/s at point A. Find the speed of the roller coaster when it reaches locations B and C....
-
Suppose the ski trail in Problem 31 is not friction less. (a) Find the work done by gravity on the skier in this case. (b) If the skier has a speed of 30 m/s at the bottom of the hill, what is the...
-
A company manufactures lawnmowers. Compute the total amount of period costs from thr following costs.
-
TestAnswerSavedHelp opens in a new windowSave & ExitSubmit Item 1 7 1 0 points Time Remaining 1 hour 2 0 minutes 1 8 seconds 0 1 : 2 0 : 1 8 Item 1 7 Time Remaining 1 hour 2 0 minutes 1 8 seconds 0 1...
-
Use the following information for the Problems below. (Algo) [The following information applies to the questions displayed below.] Lansing Company's current-year income statement and selected balance...

Study smarter with the SolutionInn App


