The following enols cannot be isolated. They rapidly tautomerize to produce ketones. In each case, draw the
Question:
(a)
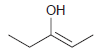
(b)
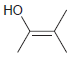
(c)
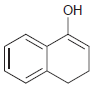
(d)
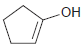
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Question Posted:





