Where might the following compounds have IRabsorptions? (b) (c) .Co (a) C -H-CH2CH CH2OH
Question:
Where might the following compounds have IRabsorptions?
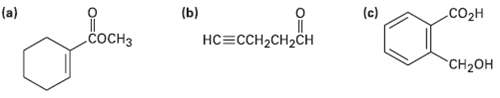
Transcribed Image Text:
(b) (c) .Coон (a) СОСНЗ нC -ссH-CH2CH CH2OH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 53% (13 reviews)
a An ester next to a double bond absorbs at 1715 cm The ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The following compounds have the same molecular formulas (C4H10). Which one would you expect to have a higher boiling point?
-
At what approximate positions might the following compounds show IRabsorptions? c??H? " alt="At what approximate positions might the following compounds show IRabsorptions? 51524" class="fr-fic...
-
At what approximate positions might the following compounds show IRabsorptions? (c) (b) CH CH (a) CH3CH2CH3 CHCCH2CH3CH2 CHCH2C3CH (f) . (d) (e) " CCH2CH2COCH CH
-
Multiple Choice 1. Which of the following concepts are pervasive in the application of generally accepted auditing standards, particularly the standards of field work and reporting? a. Internal...
-
Suppose that two firms compete in a market where consumers have identical preferences. The benefits and costs of the two firms are B 1 , C 1 and B 2 , C 2 respectively, where B 1 - C 1 > B 2 - C 2 ....
-
Easton Company included the following items in its financial statements for 2012, the current year (amounts in millions): Requirements 1. Use DuPont analysis to compute Eastons return on assets and...
-
Eulerian Use the graphs and multigraphs shown. Identify any graphs and/or multigraphs with the given characteristics. If there are none, state so. W a m n M Graph 11 Graph 12 Graph 13 Multigraph 14...
-
On January 1, Chreesh Corporation had 95,000 shares of no-par common stock issued and outstanding. The stock has a stated value of $5 per share. During the year, the following occurred. Apr. 1 Issued...
-
In which part of the purchasing process are trade shows & conferences especially helpful? None of the answers are correct Product identification and selection Analysis PO transmission All of the...
-
Core 1 Integrated Problem 8 Scenario (120 minutes) Solstice Fitness Co. (Solstice) is a privately-owned chain of high-end fitness clubs offering a range of services and amenities to its members,...
-
The IR spectrum o phenyl acetylene is shown in figure. What absorption bands can youidentify? 100 80 60 40 -C=CH 20 4000 1500 Wavenumber (cm) 3500 3000 2500 2000 1000 500 Transmittance (%)
-
Where might the following compound have IR absorptions?
-
Racially diverse nations tend to have lower levels of social support than homogenous ones. People dont feel as bound together when they are divided on ethnic lines and are less likely to embrace...
-
What role do strategic alliances, joint ventures, and collaborative partnerships play in enhancing organizational agility, accessing complementary resources and capabilities, and driving synergistic...
-
As IT consultants, you are being asked by your customer of how they would benefit using NoSQL. Can you cite two advantages of NoSQL and why there are considered as benefits.?
-
Effectively listening is a skill that most people may not realize is crucial to effective communication. IN today's age of multi-tasking,I have come to realize that oftentimes I am not fully present...
-
What is the concept analysis of long-term care in relation to Employee Records & Mandatory H.R. Documentation, Screening and background checks and Credentialing for employees?
-
How do you calculate depreciable basis? And does the base change at all depending on the depreciation method ( straight - line, activity, etc. ) ?
-
What is a local variable? How does its scope differ from that of a global variable?
-
14. In testing the existence assertion, an auditor ordinarily works from the a. Financial statements to the accounting records. b. General journal to the general ledger. c. Supporting evidence to the...
-
The vapor pressure of a 1 M ionic solution is different from the vapor pressure of a 1 M nonelectrolyte solution. In both cases, the solute is nonvolatile. Which set of diagrams best represents the...
-
Predict the major products of the following reactions. (a) 1-methylcyclohexene + aqueous Hg(OAc)2 (b) The product from part (a), treated with NaBH4 (c) 4-chlorocycloheptene + Hg(OAc)2 in CH3OH (d)...
-
Show how you would accomplish the following synthetic conversions. (a) But-1-ene 2-methoxybutane (b) 1-iodo-2-methylcyclopentane 1-methylcyclopentanol (c) 3-methylpent-1-ene 3-methylpentan-2-ol...
-
(a) Count the elements of unsaturation in the three structures shown above (parsalmide, ethynyl estradiol, and dynemicin A). (b) Draw structural formulas of at least two alkynes of each molecular...
-
The accounting records of Wall's China Shop reflected the following balances as of January 1 , Year 2 . Cash $ 1 6 , 6 0 0 Beginning inventory 1 2 , 4 6 0 ( 1 4 0 units @ $ 8 9 ) Common stock 1 5 , 8...
-
Selected balance sheet information and the income statement for Pioneer Industries for the current year are presented below. \ table [ [ Selected Balance Sheet Accounts ] , [ Accounts Receivable,$ 2...
-
Rundle Manufacturing Company established the following standard price and cost data. Sales price $ 8 . 2 0 per unit Variable manufacturing cost $ 4 . 0 0 per unit Fixed manufacturing cost $ 2 , 3 0 0...

Study smarter with the SolutionInn App


