Phosphate in urine can be determined by spectrophotometry. After removing protein from the sample, it is treated
Question:
Phosphate in urine can be determined by spectrophotometry. After removing protein from the sample, it is treated with a molybdenum compound to give, ultimately, a deep blue polymolybdate. The absorbance of the blue polymolybdate can be measured at 650 nm and is directly related to the urine phosphate concentration. A 24-hour urine sample was collected from a patient; the volume of urine was 1122 mL. The phosphate in a 1.00 mL portion of the urine sample was converted to the blue polymolybdate and diluted to 50.00 mL. A calibration curve was prepared using phosphate-containing solutions.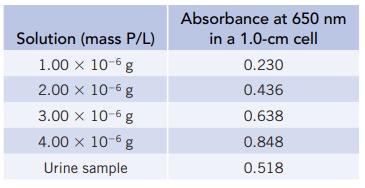
(a) What are the slope and intercept of the calibration curve?
(b) What is the mass of phosphorus per liter of urine?
(c) What mass of phosphate did the patient excrete in the one-day period?
Step by Step Answer:

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel





