Balance each of the following redox reactions in acid solution.
Question:
Balance each of the following redox reactions in acid solution.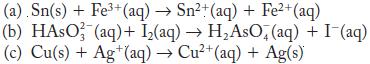
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
The process of balancing redox reactions involves breaking down the reaction into its oxidation and ...View the full answer

Answered By

Aysha Ali
my name is ayesha ali. i have done my matriculation in science topics with a+ . then i got admission in the field of computer science and technology in punjab college, lahore. i have passed my final examination of college with a+ also. after that, i got admission in the biggest university of pakistan which is university of the punjab. i am studying business and information technology in my university. i always stand first in my class. i am very brilliant client. my experts always appreciate my work. my projects are very popular in my university because i always complete my work with extreme devotion. i have a great knowledge about all major science topics. science topics always remain my favorite topics. i am also a home expert. i teach many clients at my home ranging from pre-school level to university level. my clients always show excellent result. i am expert in writing essays, reports, speeches, researches and all type of projects. i also have a vast knowledge about business, marketing, cost accounting and finance. i am also expert in making presentations on powerpoint and microsoft word. if you need any sort of help in any topic, please dont hesitate to consult with me. i will provide you the best work at a very reasonable price. i am quality oriented and i have 5 year experience in the following field.
matriculation in science topics; inter in computer science; bachelors in business and information technology
_embed src=http://www.clocklink.com/clocks/0018-orange.swf?timezone=usa_albany& width=200 height=200 wmode=transparent type=application/x-shockwave-flash_
4.40+
11+ Reviews
14+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
Balance each of the following redox reactions in acid solution.
-
Balance each of the following redox reactions in acid solution.
-
Balance each of the following redox reactions in acid solution.
-
Match the phrase that follows with the term (a-e) it describes. estimates the number of units to be manufactured to meet sales and inventory levels integrated set of operating and financing budgets...
-
After being cut from a large single-crystal boule and polished, silicon wafers undergo a high-temperature annealing process. One technique for heating the wafer is to irradiate its top surface using...
-
1. What financial risks would Mighty Leaf take by pursuing a supermarket strategy? 2. How could they moderate cash flow problems? Jill Portman and Gary Shinner associate tea with romance, relaxation,...
-
(Available-for-Sale SecuritiesStatement Presentation) Alvarez Corp. invested its excess cash in available-for-sale securities during 2006. As of December 31, 2006, the portfolio of available-for-sale...
-
Examine the bus matrix for a university shown in Figure 32.12. The university is organized as schools such as the School of Computing, School of Business Studies and each school has a portfolio of...
-
Firm Y has issued 330 million shares of stock at $1 par value and $210 million in additional paid-in capital. Retained earnings are $3.8 billion. What is the return on equity if net income is $1.5...
-
Balance each of the following redox reactions in basic solution.
-
Complete and balance each half-reaction in acid solution, and identify it as an oxidation or a reduction.
-
Suggest some ways in which seasonal changes in plant growth in Canada might affect songbirds in Mexico.
-
You are expected to suggest several functional tactics and how these short-term activities are used to achieve short term objectives and establish a competitive advantage. Within the general...
-
Carbon dioxide and nitrogen experience equimolar counterdiffusion in a circular tube whose length and diameter are 1m and 50mm, respectively. The system is at a total pressure of 1 atm and a...
-
A licensee recently was placed on court - ordered probation. Does the licensee have to report this to the Board?
-
1. Technology and Operations What task does the operations function in a manufacturing organisation and in a service organisation perform? How does operations strategy contribute to make to corporate...
-
Do the Following current market analysis - geographic , psychographic and behavioral of Klean Kanteen THIS IS THE DETAILS AND DRAFTS OF PAPER. (THIS IS THE BASIS) Open the link;...
-
A common form of elemental phosphorus is the tetrahedral P4 molecule, where all four phosphorus atoms are equivalent: At room temperature phosphorus is a solid. (a) Do you think there are any...
-
Find the market equilibrium point for the following demand and supply functions. Demand: 2p = - q + 56 Supply: 3p - q = 34
-
In our discussion of the block and tackle in Figure 3.23, we claimed that when the right end of the string is lifted through a distance L, the body of the pulley is lifted a distance L/2. Give a...
-
An astronaut measures her mass and her weight on Earth and again when she reaches the Moon. Which one of these quantities changes as a result of this trip and which one does not? Explain.
-
One end of a string is tied to the ceiling of an elevator, and the other end is tied to a rock (Fig. Q3.20). The elevator is moving in such a way that the tension in the string is zero. (a) What is...
-
A government bond matures in 30 years, makes semi-annual coupon payments of 6.0% ($120 per year) and offers a yield of 3.7% annually compounded. Assume face value is $1,000. Three years later the...
-
Your objective is: 1. Carry out a life insurance needs analysis, for each one of them (show your calculations) [30 Marks] 2. Refer to the case and the insurance plan quotes. Would you recommend...
-
TufStuff, Incorporated, sells a wide range of drums, bins, boxes, and other containers that are used in the chemical industry. One of the company s products is a heavy - duty corrosion - resistant...

Study smarter with the SolutionInn App


