Question: For each compound below, identify which position(s) is/are most likely to undergo an electrophilic aromatic substitution reaction. (a) (b) (c) (d) (e) (f) (g) (h)
For each compound below, identify which position(s) is/are most likely to undergo an electrophilic aromatic substitution reaction.
(a)
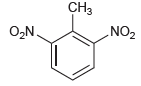
(b)
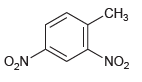
(c)
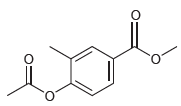
(d)
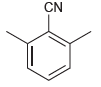
(e)
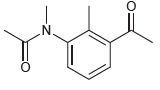
(f)
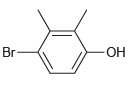
(g)
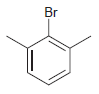
(h)
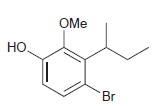
(i)
CH3 NO2 O,N. CH3 O,N NO2
Step by Step Solution
3.36 Rating (177 Votes )
There are 3 Steps involved in it
a b c... View full answer

Get step-by-step solutions from verified subject matter experts


