For each of the following complexes, determine the oxidation state of the metal and the total number
Question:
For each of the following complexes, determine the oxidation state of the metal and the total number of valence electrons it possesses.
(a)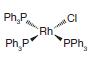
(b)
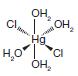
(c)
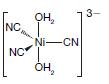
Transcribed Image Text:
Ph.P H Ph P CI PPh mee Rh
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (5 reviews)
Answer a The oxidation state of the metal in the complex is 3 a...View the full answer

Answered By

Justin Akengo
I am writing in application for the tutor position with your organisation. I am experienced in tutoring students of all abilities and I believe I am the ideal candidate for this position.
I work with students of all ages, from elementary school to college level. Whether the subject is science, Mathematics or basic study skills, I break material down into easy-to-understand concepts. In your job posting, you asked for someone who can tutor in a variety of subjects. I am comfortable explaining calculus to a college student or working with a kindergartener on spelling fundamentals.
Below are just a few core skills and qualifications I posses as a tutor;
Adept at creating study materials in a variety of academic subjects to help students improve their test scores and GPAs.
Strong interpersonal skills in working with students to help them achieve and succeed.
Have written study books adopted by a high school and a college to help students improve their skills in English and mathematics.
Have won several “Tutor of the Year” awards for work with high school and college students.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry
ISBN: 978-1118875766
12th Edition
Authors: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Question Posted:
Students also viewed these Sciences questions
-
For each of the following complexes, determine the oxidation state of the transition-metal atom. a. [CoCl(en)2(NO2)]NO2 b. [PtCl4]2 c. K3[Cr(CN)6] d. [Fe(H2O)5(OH)]2+
-
State the research problems for each of the following management decision problems. a. Should a new product be introduced? b. Should an advertising campaign that has run for three years be changed?...
-
State the appropriate nonparametric test for each of the following examples. (a) A physiological psychologist compares differences in the amount of chemical activity in a certain brain region between...
-
1. Prepare a schedule of cost of goods manufactured for Denim Bones for the year ended December 31, 2024. 2. Prepare an income statement for Denim Bones for the year ended December 31, 2024. 3. How...
-
The percentages of Americans who are satisfied with the way things are in the United States are shown in Table 59 for various years. Let p be the percentage of Americans at t years since 1990 who are...
-
For the same system as described in problem 5, complete the following. (a) The initial displacements for the system's free vibration are \(x_{1}(0)=0.378\) \(\mathrm{mm}\) and \(x_{2}(0)=1...
-
What must be included in a motion for summary judgment?
-
Narcisco Publications established the following standard price and costs for a hardcover picture book that the company produces: Standard price and variable costs Sales price...
-
SQL trigger, that should be named BI_FILM_LANG, to append text to the description of every new film inserted into the database. It is based on the language (language_id) and the original language...
-
Assume a TCP server expects to receive byte 2001, but it receives a segment with sequence number 2200. What is the reaction of the TCP server to this event? Can you justify the reaction?
-
What is the product of the following SuzukiMiyaura coupling? Br B(OR)2 + Pd catalyst Base
-
In The Chemistry of Complex Cross Couplings we showed a Heck reaction used in the synthesis of ecteinascidin 743, a natural compound with anticancer activity isolated from the marine tunicate...
-
If the resultant couple of the two couples acting on the fire hydrant is MR = {−15i + 30j} N m, determine the force magnitude P. Given: a = 0.2 m b = 0.150 m M = (-15 30 0) N m F = 75 N Pi Fj...
-
Present Value Computations Using the present value tables, solve the following. Round your answers to two decimal places. Required: 1. What is the present value on January 1, 2016, of $30,000 due on...
-
KOJOKARO This exercise is a review of the setup of a company. The account structure (i.e Chart of Accounts) was created by an employee of Kojokaro. Account classes have not been shown in the...
-
Consider the simple regression model Y =B + Bx +4 t = 1,2, ...,T where u, pu, + & with lpl < 1 and E(e) = 0, E(?) = o?, E(EE) = 0,t #s a. What are the consequences of having first order...
-
196. By which process man has obtained many breeds of plants and animals for agriculture, horticulture, food and security? (1) Natural selection (2) Random breeding (3) Artificial selection (4) Both...
-
An m = 0.5 kg compresses a spring of force constant 500 N/m a distance 0.2 m from its natural length. When the block is released, it travels up a rough incline that is h = 0.4 m high and 2 m long....
-
Which store do you think is more expensive-physical or online? A recent survey (USA Today, December 10, 2012, p. 1B) found that 46% of people aged 20 to 40 thought that physical stores were more...
-
Which property determines whether a control is available to the user during run time? a. Available b. Enabled c. Unavailable d. Disabled
-
Predict the positions of the major absorption bands in the IR spectra of thesecompounds: CH30 CH-NH, a) CH3CH=CHCCH; c) b) C-H NO, e) CH-CH,C3- d) CH,CH,CH,OH CH3
-
Explain how IRD spectroscopy could be used to distinguish between these compounds: CH;CH,C=CH a) CH,CH,CH=CH; and CH3 H- b) CH, nd CH3 CCH3 CH c) and d) CH.CH,CH,C,NH, and CH.CH,NHCH,CH,
-
List the positions of the important absorption bands in the IR spectra of these compounds: b) H,H-CH-C3DN ) -H,H,NH> CCH,CH3 c) CH;CH,COCH;CH,CH3 d)
-
Townsend Produce agrees to sell 100 crates of lettuce to Metro Grocers under a shipment contract. Townsend arranges for Excel Transport to pick up and deliver the lettuce, which Excel Transport does....
-
Assume an organic compound has a partition coefficient between water and ethyl acetate equal to 8.12. If there are initially 7.10 grams of the compound dissolved in 75.0 mL of water, how many grams...
-
NO. DATE Use the table below Write a Java program that will display the output below. code may vary... For your reference QUANTITY ITEM PRICE 1 Bond Paper (rim) 230 1 Notebook 30 1 1 Printer Ink 5500...

Study smarter with the SolutionInn App


