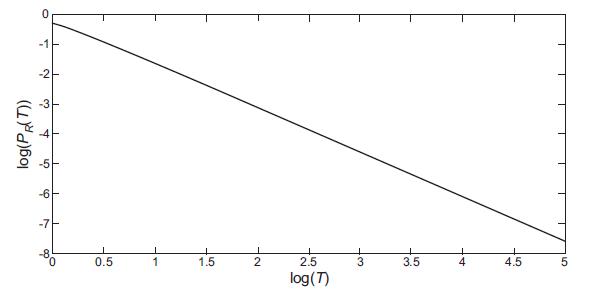
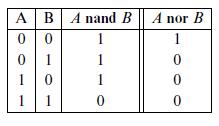
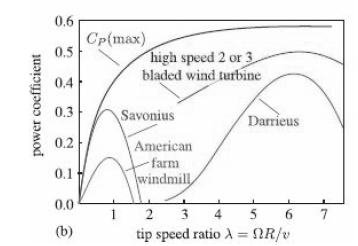
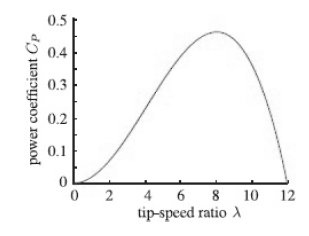
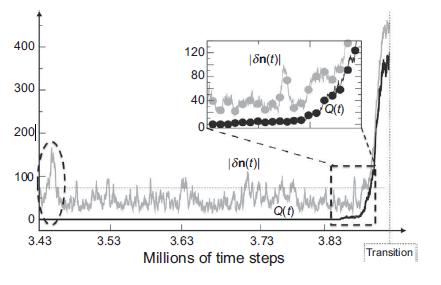
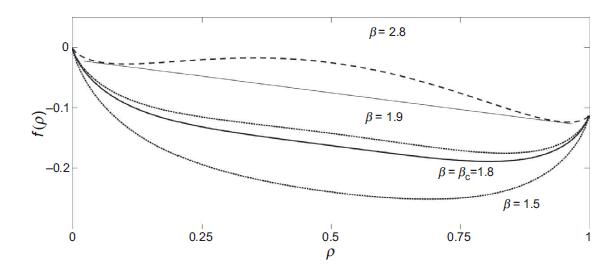
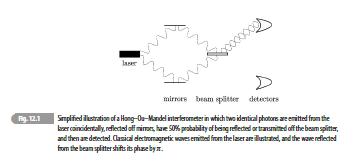
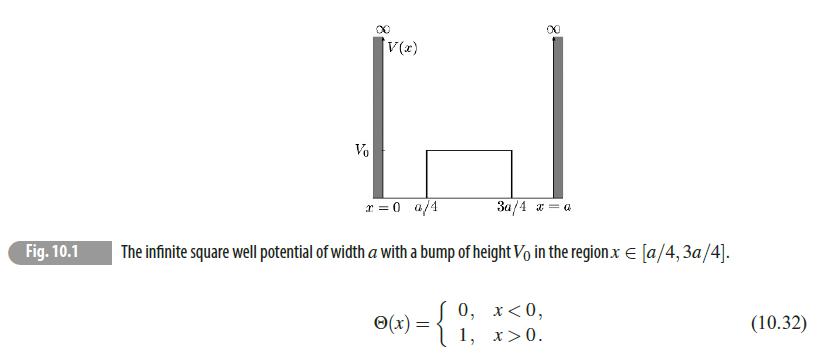
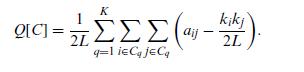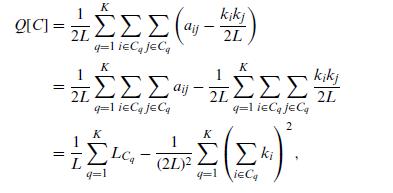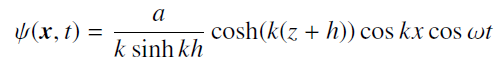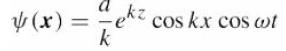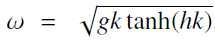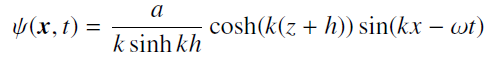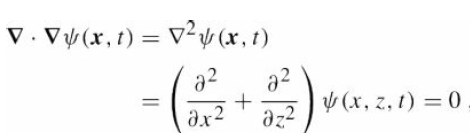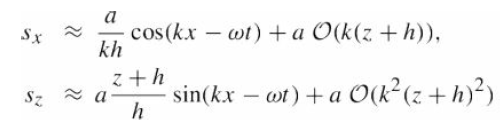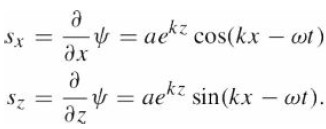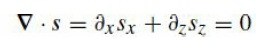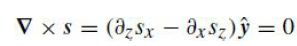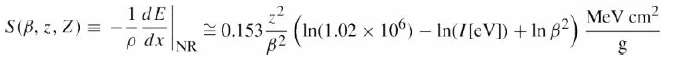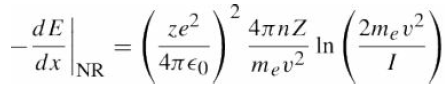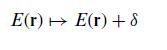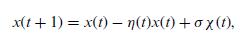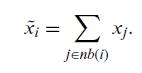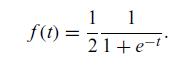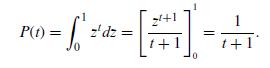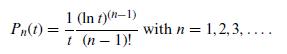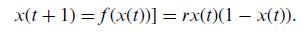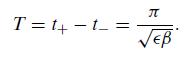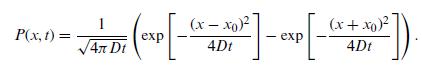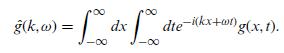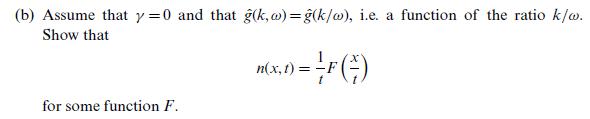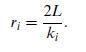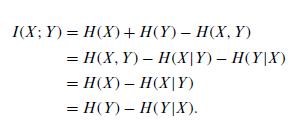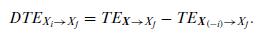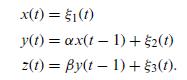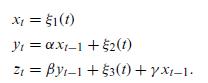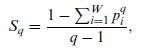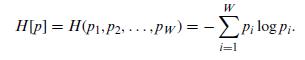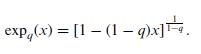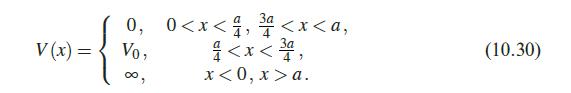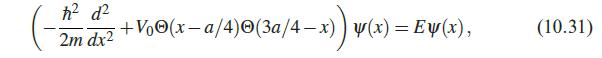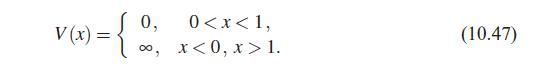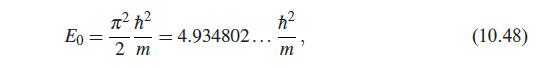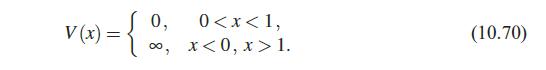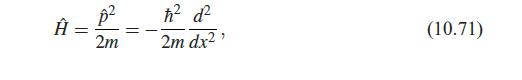Quantum Mechanics A Mathematical Introduction 1st Edition Andrew J. Larkoski - Solutions
Unlock the secrets of quantum mechanics with our comprehensive resource for "Quantum Mechanics A Mathematical Introduction" by Andrew J. Larkoski. Dive into expertly crafted solutions and an extensive answers key, all available online. Our solution manual offers step-by-step answers to complex problems, ensuring a clear understanding of each concept. Access a free download of the solutions PDF, featuring solved problems and chapter solutions tailored to enrich your learning experience. Whether you seek a test bank or an instructor manual, our textbook resources provide all the questions and answers you need.
![]()
![]() New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
![]()
![]()