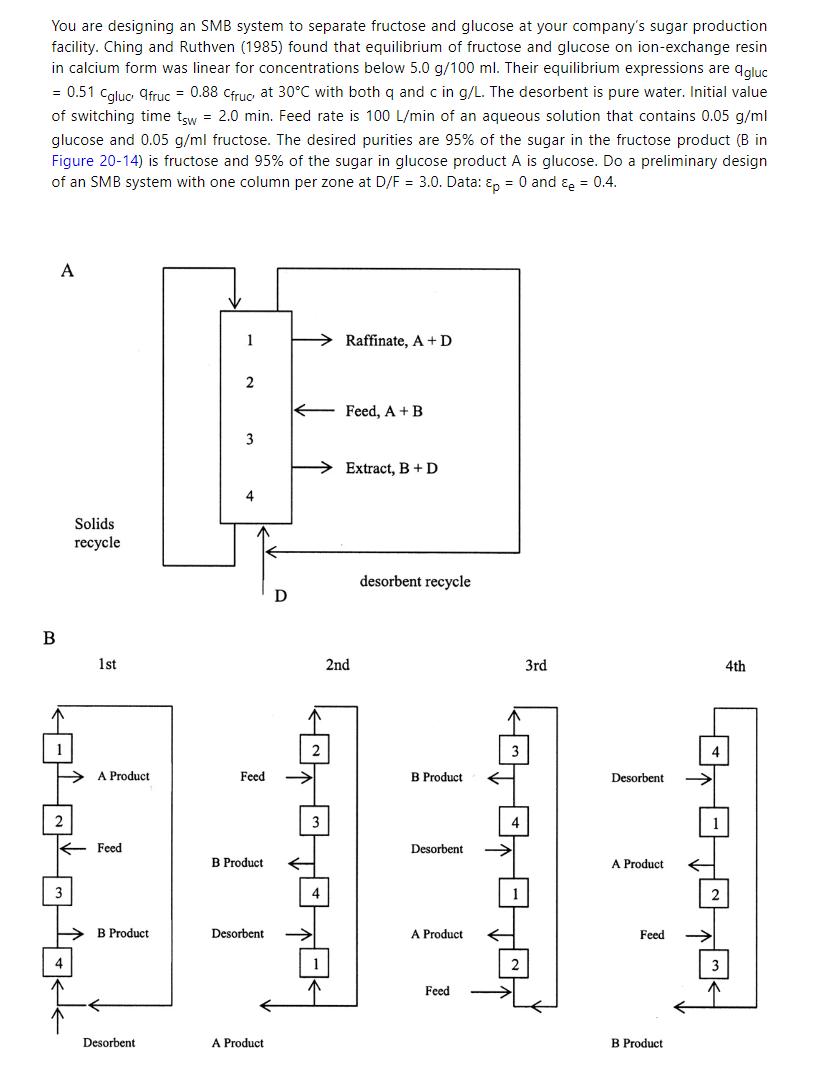
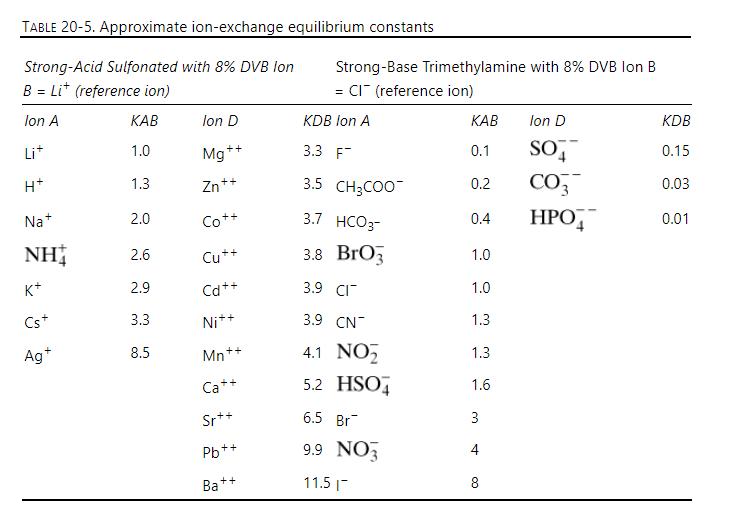
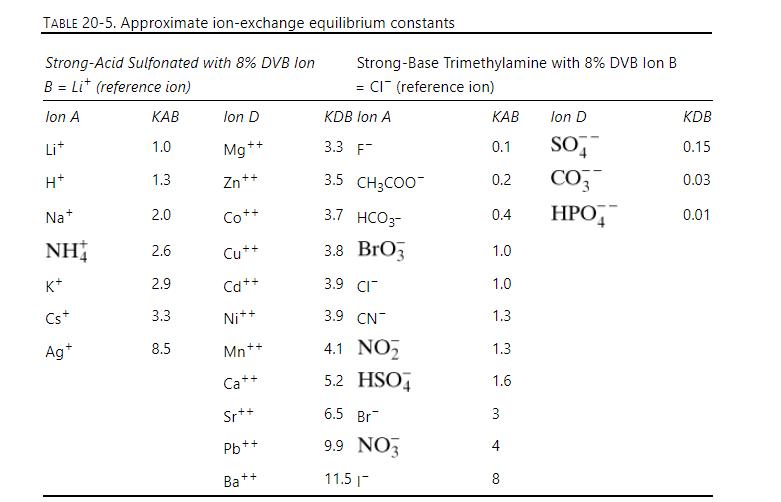
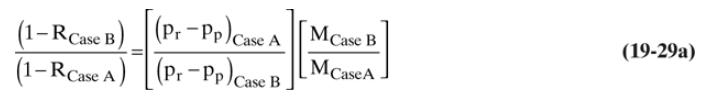
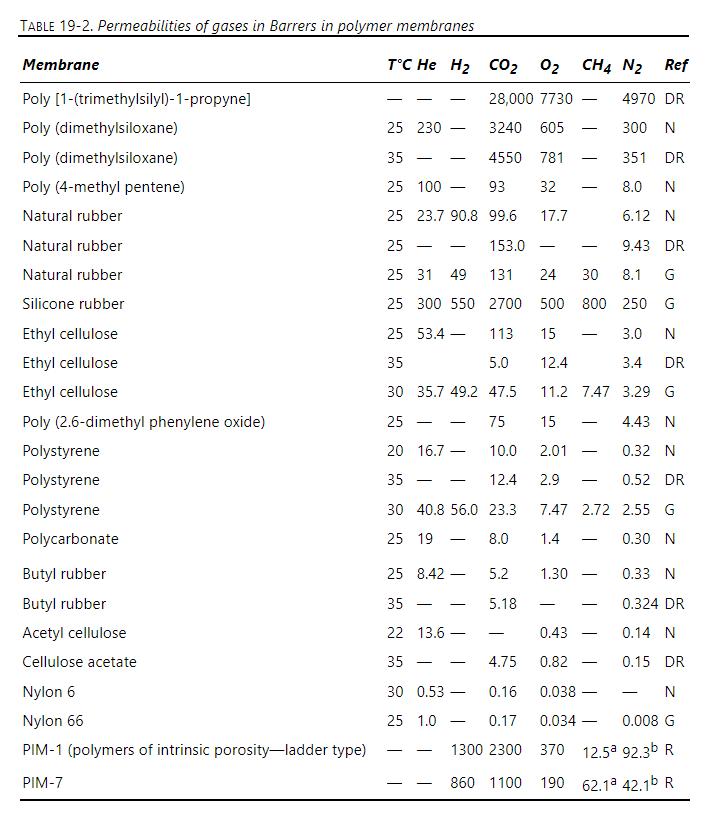
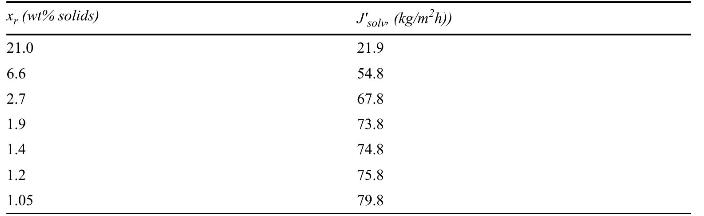
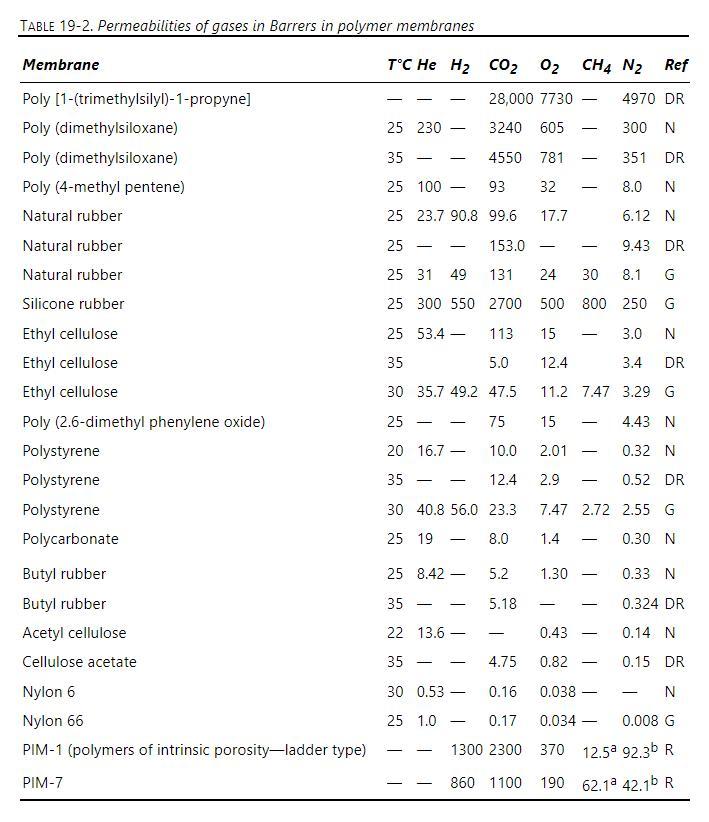
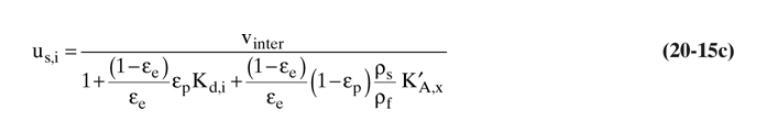Separation Process Engineering Includes Mass Transfer Analysis 5th Edition Phillip Wankat - Solutions
Discover comprehensive solutions for "Separation Process Engineering Includes Mass Transfer Analysis 5th Edition" by Phillip Wankat. Access our online answers key and solution manual to tackle complex problems efficiently. Our solutions PDF offers step-by-step answers to all textbook questions, providing you with a reliable test bank for academic success. Explore chapter solutions and solved problems with ease. Whether you're seeking an instructor manual or a free download of expert solutions, our resources support your learning journey. Enhance your understanding with detailed questions and answers, designed to simplify your study process.
![]()
![]() New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
New Semester Started
Get 50% OFF
Study Help!
--h --m --s
Claim Now
![]()
![]()